What Is Cellular Senescence?
Cellular senescence is a biological stress response in which damaged or stressed cells permanently stop dividing but do not die. These cells remain metabolically active and continue to interact with their environment.
Senescence is triggered by multiple forms of cellular damage, including:
- DNA damage
- Oxidative stress (reactive oxygen species)
- Telomere shortening
- Oncogene activation
- Chemotherapy and radiation exposure
At first, this process acts as a protective mechanism. By halting cell division, senescence prevents damaged cells from becoming cancerous. However, over time, senescent cells accumulate and begin to promote inflammation, metabolic dysfunction, and tumor progression.
This dual role makes cellular senescence one of the most complex processes in cancer biology.
The Dual Role of Senescence in Cancer
Tumor Suppression (Early Phase)
In early stages, senescence acts as a powerful anti-cancer mechanism.
When cells experience stress or DNA damage, key tumor suppressor pathways activate:
- p53 pathway
- p16INK4a pathway
- Retinoblastoma (RB) signaling
These pathways enforce permanent cell cycle arrest. The damaged cell can no longer divide, preventing the spread of mutations.
This is particularly important during:
- Early tumor formation
- Pre-cancerous lesions
- DNA damage response after therapy
In this phase, senescence functions as a barrier against cancer development.
Tumor Promotion (Late Phase)
Over time, senescent cells become harmful.
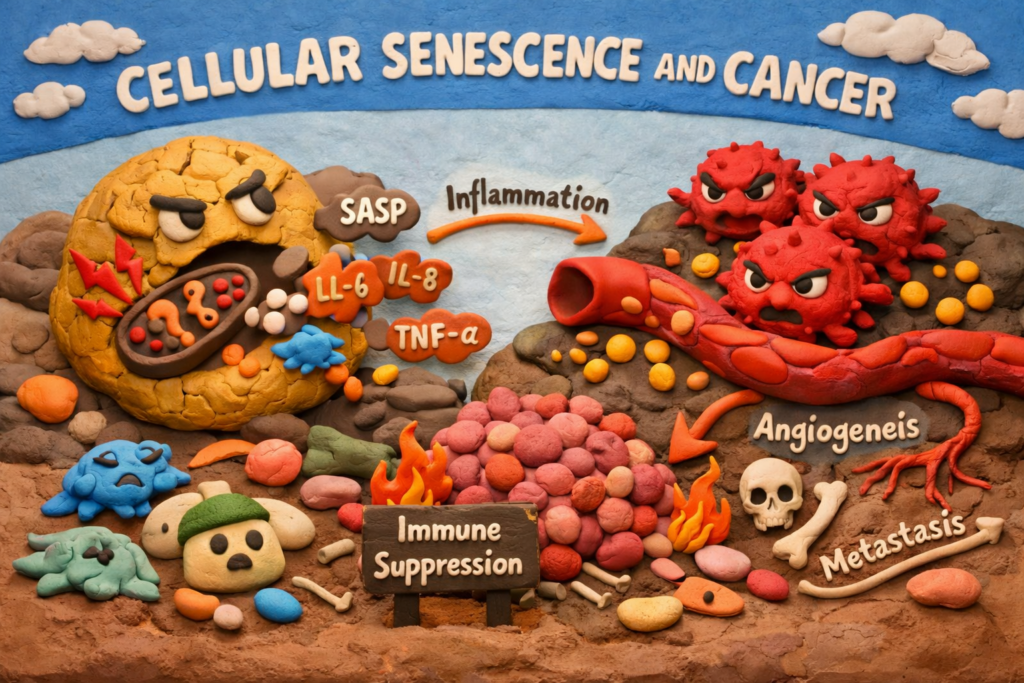
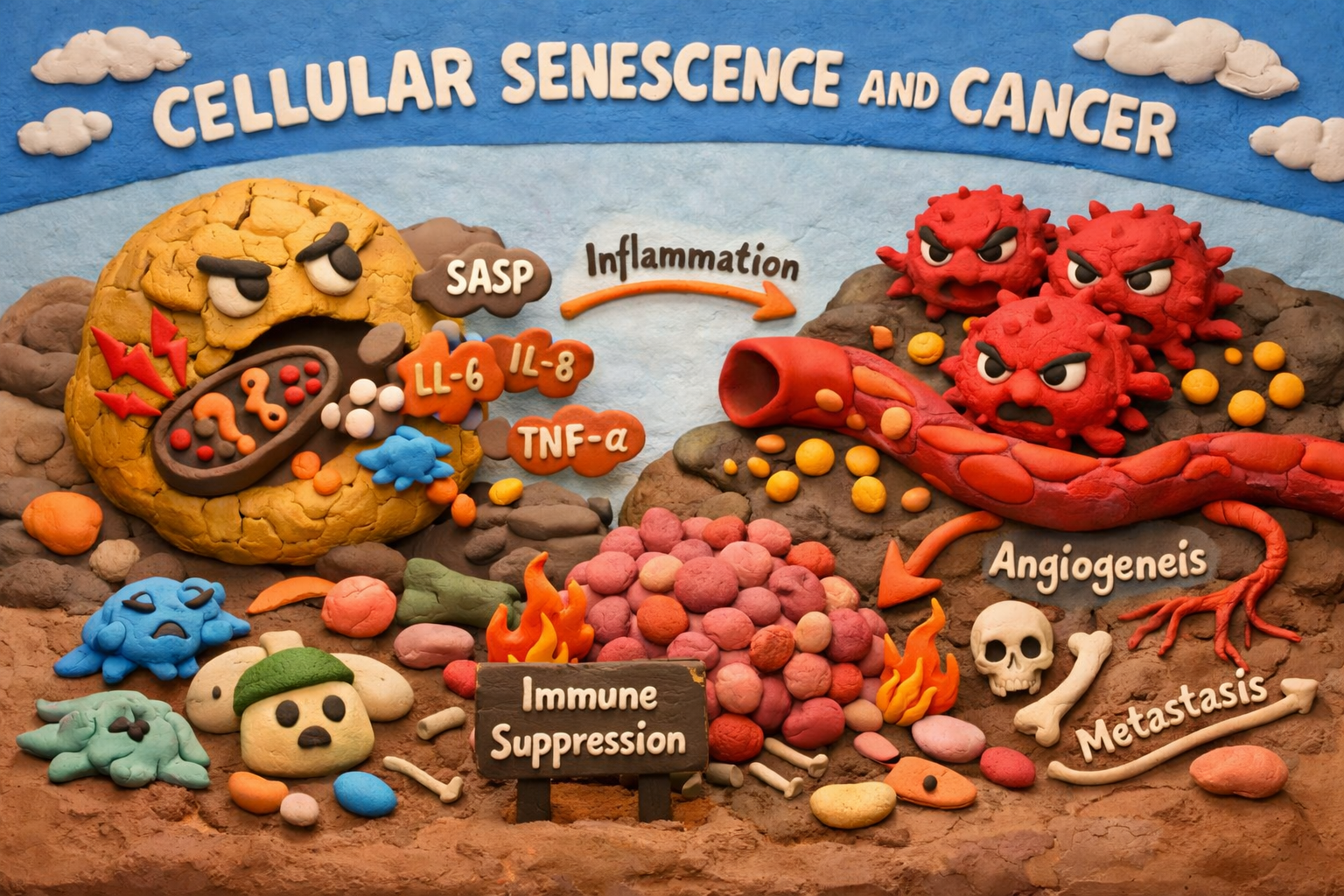
Instead of remaining silent, they begin secreting a mix of signaling molecules known as the Senescence-Associated Secretory Phenotype (SASP).
This includes:
- Pro-inflammatory cytokines (IL-6, IL-8)
- Growth factors
- Proteases that remodel tissue
- Immune-modulating signals
These secretions create a pro-tumor environment.
Key effects include:
- Chronic inflammation
- Stimulation of nearby cancer cell growth
- Increased angiogenesis
- Immune suppression
- Promotion of metastasis
This shift transforms senescence from a protective process into a driver of tumor survival and recurrence.
The Senescence-Associated Secretory Phenotype (SASP)
SASP is the primary reason senescent cells become dangerous in cancer.
Instead of remaining inactive, senescent cells actively reshape their environment.
Key SASP Components
- Cytokines: IL-1, IL-6, TNF-alpha
- Chemokines: Recruit immune cells but often in dysfunctional ways
- Growth factors: VEGF, TGF-beta
- Matrix remodeling enzymes: MMPs
These signals create a microenvironment that favors tumor growth.
SASP and Tumor Microenvironment
The tumor microenvironment is heavily influenced by SASP signaling.
SASP contributes to:
- Persistent inflammation
- Breakdown of extracellular matrix
- Recruitment of immunosuppressive cells
- Activation of survival pathways like NF-κB and STAT3
This environment allows cancer cells to thrive even under stress.
External reference: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3531129/
Senescence, ROS, and Oxidative Stress
Reactive oxygen species (ROS) are central to both the induction and consequences of senescence.
ROS as a Trigger
High levels of ROS damage DNA, proteins, and lipids. This damage activates senescence pathways.
Sources of ROS include:
- Mitochondrial dysfunction
- Chemotherapy and radiation
- Chronic inflammation
- Metabolic stress
ROS-induced DNA damage is one of the most common triggers of senescence.
ROS in Senescent Cells
Once cells become senescent, they continue producing elevated ROS.
This creates a feedback loop:
- ROS → DNA damage → senescence
- Senescence → mitochondrial dysfunction → more ROS
This cycle drives:
- Chronic inflammation
- Tissue damage
- Tumor-promoting signaling
External reference: https://www.nature.com/articles/nrm.2016.85
Mitochondrial Dysfunction in Senescent Cells
Senescent cells show profound metabolic changes, especially in mitochondria.
Key Mitochondrial Changes
- Reduced ATP efficiency
- Increased ROS production
- Altered mitochondrial dynamics (fusion/fission imbalance)
- Impaired mitophagy (damaged mitochondria are not cleared)
These changes push cells toward a dysfunctional metabolic state.
Metabolic Reprogramming
Senescent cells shift metabolism to support SASP production.
This includes:
- Increased glycolysis
- Altered NAD+/NADH balance
- Enhanced inflammatory signaling
The metabolic burden of SASP secretion further stresses mitochondria, amplifying dysfunction.
Internal link: https://helping4cancer.com/cancer-and-mitochondria/
Senescence and Cancer Treatment
Cancer therapies often induce senescence intentionally or unintentionally.
Therapy-Induced Senescence (TIS)
Chemotherapy and radiation can push cancer cells into senescence rather than killing them.
This has mixed outcomes:
Benefits:
- Stops tumor cell proliferation
- Reduces tumor size temporarily
Risks:
- Senescent cancer cells remain alive
- SASP promotes relapse
- Resistant cells may re-enter the cell cycle
This explains why tumors can shrink initially but later return.
Senescence and Recurrence
Senescent cells contribute to cancer recurrence through:
- Reactivation of dormant tumor cells
- Promotion of stem-like cancer cells
- Protection of tumor niches
These mechanisms allow cancer to survive treatment and re-emerge later.
External reference: https://pubmed.ncbi.nlm.nih.gov/25959794/
Senescence and Immune System Interactions
The immune system plays a critical role in clearing senescent cells.
Immune Clearance
Healthy immune function can remove senescent cells through:
- Natural killer (NK) cells
- Cytotoxic T cells
- Macrophages
This process is essential for preventing accumulation.
Immune Evasion
However, senescent cells can evade immune detection.
They do this by:
- Altering surface markers
- Secreting immunosuppressive factors
- Recruiting regulatory immune cells
This allows them to persist and contribute to tumor progression.
Internal link: https://helping4cancer.com/tumor-immune-cloaking/
Senescence in the Tumor Microenvironment
Senescence is not limited to cancer cells. It also affects surrounding cells.
Affected Cell Types
- Fibroblasts
- Immune cells
- Endothelial cells
When these cells become senescent, they contribute to a pro-tumor environment.
Effects on Tumor Growth
Senescent stromal cells:
- Support tumor metabolism
- Enhance angiogenesis
- Promote invasion and metastasis
This creates a cooperative environment where cancer cells benefit from surrounding dysfunction.
Internal link: https://helping4cancer.com/tumor-survival-network/
Senescence, Aging, and Cancer Risk
Senescent cells accumulate naturally with age.
This contributes to:
- Chronic inflammation (inflammaging)
- Declining immune function
- Increased cancer risk
Older tissues often contain higher levels of senescent cells, creating a favorable environment for tumor development.
Targeting Senescence in Cancer Therapy
Understanding senescence has led to new therapeutic strategies.
Senolytics
Senolytics are compounds designed to eliminate senescent cells.
Potential benefits include:
- Reducing inflammation
- Preventing tumor recurrence
- Improving tissue health
Senomorphics
Senomorphics aim to suppress SASP without killing the cell.
They work by:
- Inhibiting inflammatory signaling
- Reducing cytokine release
- Modulating metabolic pathways
Metabolic Targeting
Because senescent cells rely on altered metabolism, targeting:
- Mitochondrial dysfunction
- ROS balance
- Energy pathways
may disrupt their survival.
Internal link: https://helping4cancer.com/cancer-oxidative-stress/
Why Cellular Senescence Matters in Cancer
Cellular senescence sits at the intersection of:
- DNA damage response
- Metabolic dysfunction
- Immune regulation
- Tumor microenvironment signaling
It explains several critical cancer behaviors:
- Why tumors initially respond to treatment
- Why inflammation persists after therapy
- Why recurrence occurs months or years later
Key Takeaways
- Cellular senescence stops damaged cells from dividing but does not eliminate them
- Early senescence suppresses tumors, but long-term accumulation promotes cancer
- SASP drives inflammation, tumor growth, and immune evasion
- ROS and mitochondrial dysfunction reinforce senescence and cancer survival
- Therapy-induced senescence may contribute to recurrence
- Targeting senescent cells is an emerging strategy in cancer treatment
External References
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3531129/
- https://www.nature.com/articles/nrm.2016.85
- https://pubmed.ncbi.nlm.nih.gov/25959794/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6351807/
- https://www.nature.com/articles/s41568-020-0261-2
Internal Links
- https://helping4cancer.com/cancer-and-mitochondria/
- https://helping4cancer.com/cancer-oxidative-stress/
- https://helping4cancer.com/tumor-survival-network/
- https://helping4cancer.com/tumor-immune-cloaking/
- https://helping4cancer.com/cancer-cell-cycle/
Table of Contents