What Is Fatty Acid Oxidation?
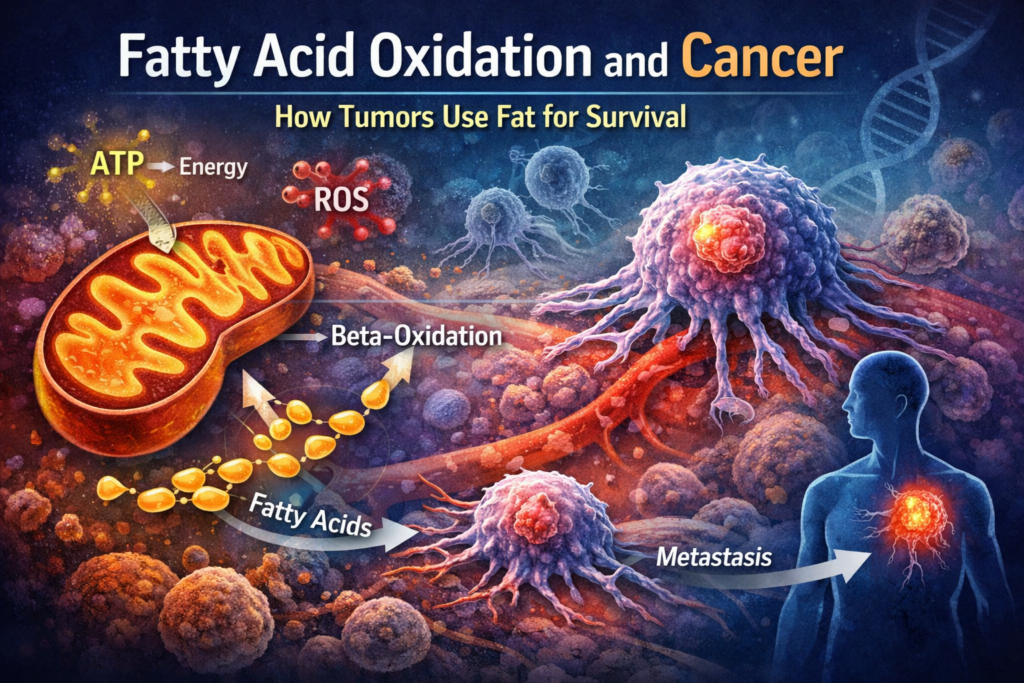
Fatty acid oxidation (FAO), also known as beta-oxidation, is a metabolic pathway that breaks down fatty acids into smaller units to produce energy. This process occurs primarily in the mitochondria and generates large amounts of ATP, NADH, and FADH2.
Under normal conditions, FAO is used during fasting, prolonged exercise, or carbohydrate restriction. It allows cells to switch from glucose dependence to fat-based energy production.
In cancer, however, this pathway can be hijacked. Certain tumors activate FAO to survive under metabolic stress, especially when glucose is limited or when they are exposed to therapies that disrupt glycolysis.
Why Fatty Acid Oxidation Matters in Cancer
Cancer cells are metabolically flexible. While many tumors rely heavily on glycolysis (the Warburg effect), others can shift toward FAO when needed.
This metabolic switch provides several advantages:
- Sustained energy during nutrient deprivation
- Resistance to metabolic therapies
- Protection against oxidative stress
- Support for metastasis and tumor spread
FAO becomes particularly important in harsh tumor environments where oxygen, glucose, and nutrients are limited.
Tumors that rely on FAO are often more aggressive and harder to treat because they can adapt to multiple metabolic conditions.
The FAO Pathway: Step-by-Step
Fatty acid oxidation is a tightly regulated mitochondrial process involving multiple steps:
Fatty Acid Uptake and Activation
Fatty acids enter the cell through transporters such as CD36 and FATP proteins. Once inside, they are converted into fatty acyl-CoA molecules.
Transport into the Mitochondria
Long-chain fatty acids cannot directly cross the mitochondrial membrane. They require a transport system known as the carnitine shuttle.
The key enzyme in this process is CPT1 (carnitine palmitoyltransferase 1), which regulates the entry of fatty acids into mitochondria.
CPT1 is often upregulated in cancer cells that depend on FAO.
Beta-Oxidation Cycle
Inside the mitochondria, fatty acids are broken down through repeated cycles. Each cycle produces:
- Acetyl-CoA
- NADH
- FADH2
Acetyl-CoA enters the TCA cycle, while NADH and FADH2 feed into the electron transport chain to generate ATP.
This makes FAO one of the most energy-efficient pathways available to cancer cells.
FAO vs Glycolysis: A Metabolic Tug-of-War
Cancer metabolism is not fixed. Many tumors can switch between glycolysis and FAO depending on environmental conditions.
Glycolysis provides rapid energy and supports biosynthesis. FAO, on the other hand, provides sustained, high-efficiency energy.
Key differences include:
- Glycolysis is fast but inefficient
- FAO is slower but produces more ATP
- Glycolysis dominates in oxygen-poor environments
- FAO supports long-term survival and stress resistance
Some tumors use both pathways simultaneously, creating a hybrid metabolic phenotype that enhances survival.
FAO and Mitochondrial Function
Fatty acid oxidation is tightly linked to mitochondrial health and function. Tumors that rely on FAO often have highly active mitochondria.
This allows them to:
- Maintain ATP production under stress
- Regulate redox balance
- Control reactive oxygen species (ROS) levels
FAO can both generate and buffer oxidative stress depending on the context.
By producing NADH and FADH2, FAO feeds electrons into the electron transport chain, which can increase ROS production. However, it also supports antioxidant systems that help cancer cells manage oxidative damage.
This balance is critical for tumor survival.
FAO and Cancer Stem Cells
Cancer stem cells are a small subpopulation of tumor cells that drive recurrence, resistance, and metastasis.
These cells often rely heavily on FAO rather than glycolysis.
FAO supports cancer stem cells by:
- Providing long-lasting energy
- Enhancing resistance to chemotherapy
- Supporting self-renewal and survival pathways
Studies have shown that inhibiting FAO can reduce cancer stem cell populations and increase sensitivity to treatment.
This makes FAO a key target in efforts to prevent relapse.
FAO and Metastasis
Metastatic cancer cells must survive in circulation, invade new tissues, and adapt to unfamiliar environments.
FAO plays a central role in this process.
It provides:
- Energy for migration and invasion
- Protection against oxidative stress during detachment
- Adaptability in nutrient-poor environments
For example, ovarian and breast cancer cells have been shown to increase FAO during metastasis.
Adipocyte-rich environments, such as the omentum or bone marrow, can supply fatty acids that fuel tumor growth through FAO.
FAO and the Tumor Microenvironment
The tumor microenvironment is often low in glucose but rich in lipids, especially in tissues like adipose tissue.
Cancer cells can take advantage of this by increasing FAO.
Interactions between cancer cells and surrounding fat cells can further enhance this process:
- Adipocytes release fatty acids
- Cancer cells uptake these fatty acids
- FAO is activated to generate energy
This metabolic cooperation allows tumors to thrive even when traditional energy sources are scarce.
FAO and Therapy Resistance
One of the most important roles of FAO in cancer is its contribution to therapy resistance.
When treatments target glycolysis or rapidly dividing cells, tumors can shift toward FAO to survive.
FAO contributes to resistance by:
- Maintaining ATP levels under treatment stress
- Reducing oxidative damage
- Supporting DNA repair mechanisms
- Enhancing survival signaling pathways
This metabolic flexibility makes cancer more difficult to eliminate.
Targeting FAO alongside other pathways is an area of active research.
Key Enzymes and Targets in FAO
Several enzymes regulate fatty acid oxidation and are commonly altered in cancer:
CPT1 (Carnitine Palmitoyltransferase 1)
CPT1 controls the rate-limiting step of FAO. It is frequently overexpressed in cancers that rely on fat metabolism.
ACADs (Acyl-CoA Dehydrogenases)
These enzymes catalyze the first step of beta-oxidation and are essential for fatty acid breakdown.
PPAR Signaling
Peroxisome proliferator-activated receptors regulate genes involved in lipid metabolism and FAO.
Activation of PPAR pathways can enhance FAO and support tumor survival.
FAO and Fasting: A Metabolic Intersection
Fasting shifts the body toward fat utilization and increases fatty acid oxidation.
While this can stress certain cancer cells, it may also benefit tumors that are adapted to FAO.
In FAO-dependent cancers, fasting can:
- Provide additional fatty acids
- Enhance mitochondrial energy production
- Support tumor survival under stress
This highlights the importance of understanding tumor metabolism before applying metabolic interventions.
Not all tumors respond the same way to fasting or dietary changes.
FAO and Reactive Oxygen Species (ROS)
Fatty acid oxidation influences reactive oxygen species in complex ways.
On one hand, increased electron transport chain activity can elevate ROS levels.
On the other hand, FAO supports antioxidant defenses that neutralize ROS.
Cancer cells use this balance to their advantage:
- Moderate ROS levels promote signaling and growth
- Excess ROS is buffered to prevent cell death
FAO helps maintain this optimal redox state.
Disrupting FAO can push ROS beyond tolerable levels, leading to cancer cell death.
Therapeutic Strategies Targeting FAO
Because of its role in survival and resistance, FAO is an attractive target for cancer therapy.
Strategies under investigation include:
- CPT1 inhibitors to block fatty acid entry into mitochondria
- Drugs that disrupt mitochondrial metabolism
- Combination therapies targeting both glycolysis and FAO
- Approaches that increase ROS while inhibiting FAO defenses
These strategies aim to remove the metabolic flexibility that allows cancer to survive.
FAO in Different Cancer Types
Fatty acid oxidation is not equally important in all cancers.
It is particularly relevant in:
- Leukemia and lymphoma
- Prostate cancer
- Breast cancer
- Ovarian cancer
- Glioblastoma
These cancers often show increased expression of FAO-related enzymes and pathways.
Understanding the metabolic profile of a tumor is essential for targeting it effectively.
Why Fatty Acid Oxidation Is a Critical Survival Pathway
Fatty acid oxidation represents a powerful survival mechanism for cancer cells.
It allows tumors to:
- Adapt to nutrient scarcity
- Resist treatment
- Maintain energy production under stress
- Support metastasis and recurrence
Rather than relying on a single pathway, cancer uses a network of metabolic options.
FAO is one of the most important components of this network.
External References
National Cancer Institute – https://www.cancer.gov
PubMed – https://pubmed.ncbi.nlm.nih.gov
Nature Reviews Cancer – https://www.nature.com/nrc
Cell Metabolism Journal – https://www.cell.com/cell-metabolism
NIH – https://www.nih.gov
Internal Links
https://helping4cancer.com/cancer-metabolism/
https://helping4cancer.com/cancer-oxidative-stress/
https://helping4cancer.com/ampk-cancer/
https://helping4cancer.com/mitochondria-cancer/
https://helping4cancer.com/warburg-effect-cancer/
Table of Contents