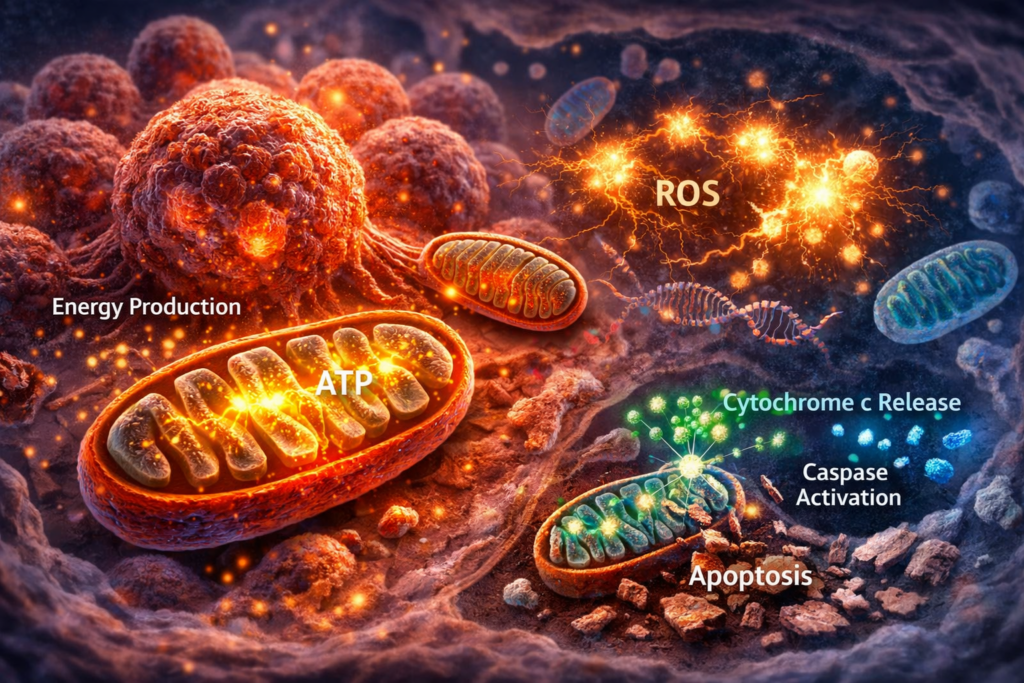
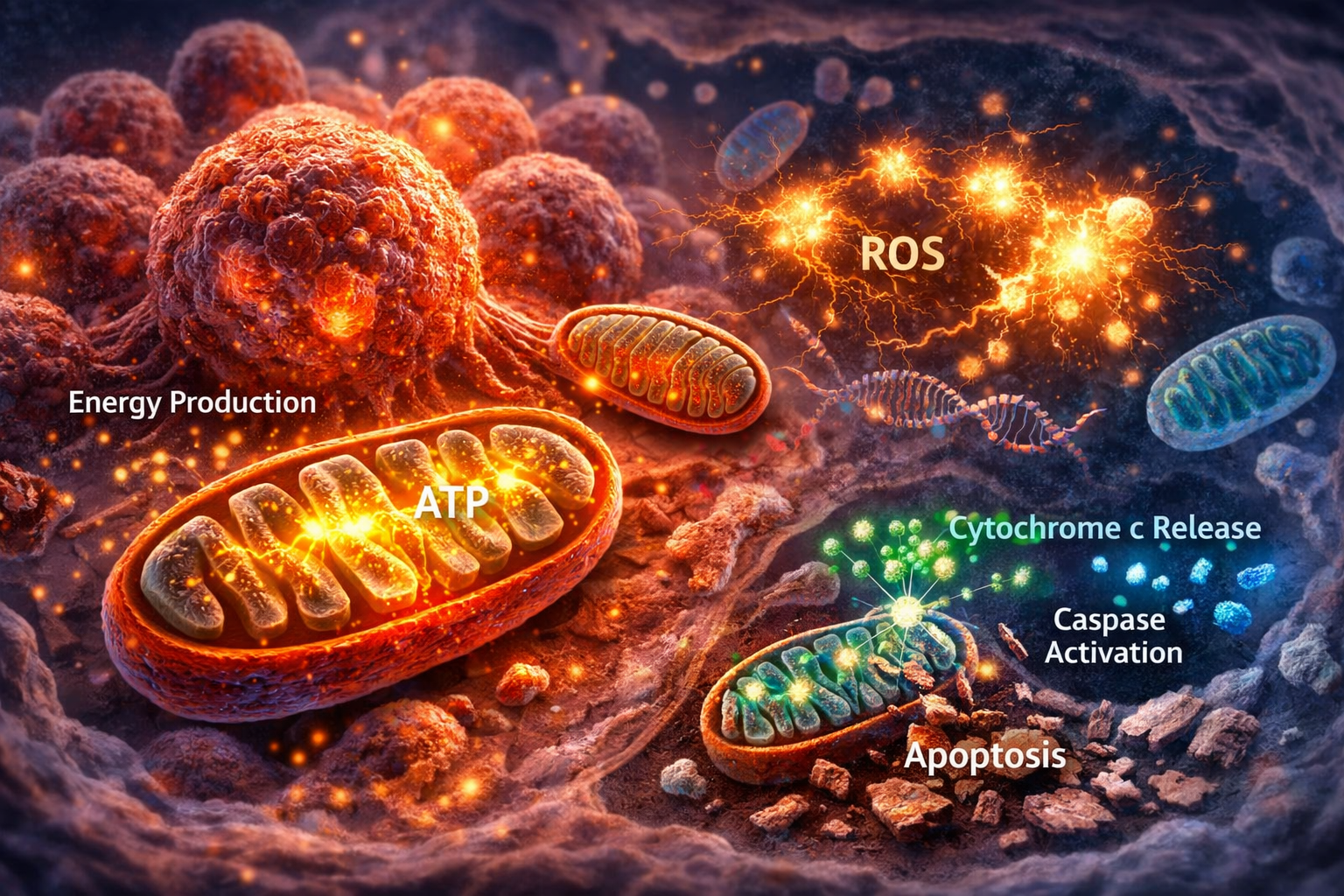
Mitochondria are often called the “power plants” of the cell because they generate energy in the form of ATP. In healthy cells, mitochondria play a central role in metabolism, signaling, and programmed cell death. However, in cancer, mitochondria are reprogrammed to support tumor growth, survival, and resistance to treatment.
Understanding how mitochondria function inside cancer cells is critical because they sit at the center of three key processes: energy production, reactive oxygen species (ROS) generation, and apoptosis (cell death). These functions make mitochondria one of the most important targets in modern cancer research.
This guide explains how mitochondria behave differently in cancer cells and why targeting mitochondrial pathways may help disrupt tumor survival.
What Are Mitochondria?
Mitochondria are specialized structures inside cells responsible for producing energy through a process called oxidative phosphorylation. They convert nutrients such as glucose and fatty acids into ATP, which powers cellular activity.
In addition to energy production, mitochondria regulate:
- Reactive oxygen species (ROS)
- Calcium signaling
- Cell death (apoptosis)
- Metabolic flexibility
In normal cells, mitochondrial activity is tightly regulated. In cancer cells, however, these processes become altered to favor growth and survival.
Mitochondrial Metabolism in Cancer
Cancer cells are known for metabolic reprogramming. While many tumors rely heavily on glycolysis (the Warburg effect), mitochondria remain active and essential.
Metabolic Flexibility
Cancer cells can switch between energy sources depending on their environment. Mitochondria allow tumors to:
- Use glucose, glutamine, and fatty acids
- Adapt to low oxygen conditions
- Survive nutrient deprivation
This flexibility gives cancer cells a survival advantage, especially during treatment.
Increased Biosynthesis
Mitochondria provide more than just energy. They generate building blocks needed for rapid cell division, including:
- Lipids for cell membranes
- Amino acids for protein synthesis
- Nucleotides for DNA replication
These biosynthetic functions are critical for tumor expansion.
Link to Tumor Metabolism
For a deeper understanding of how cancer reprograms energy systems, see:
Tumor Metabolic Switch → https://helping4cancer.com/tumor-metabolic-switch/
Tumor Metabolism vs Normal Cells → https://helping4cancer.com/tumor-metabolism-vs-normal-cells/
Reactive Oxygen Species (ROS) and Cancer
Mitochondria are the primary source of reactive oxygen species (ROS) in cells. ROS are highly reactive molecules that can damage DNA, proteins, and lipids.
ROS as a Double-Edged Sword
In cancer, ROS play a complex role:
- Moderate ROS levels promote tumor growth and signaling
- High ROS levels can damage and kill cancer cells
Cancer cells carefully balance ROS levels to maximize growth while avoiding cell death.
ROS and DNA Damage
Elevated ROS can lead to:
- DNA mutations
- Genomic instability
- Activation of oncogenes
These changes contribute to cancer progression and diversity within tumors.
ROS and Therapy
Many treatments, including chemotherapy and radiation, work by increasing ROS to toxic levels. When ROS overwhelm the cancer cell, it leads to cell death.
Related topic:
Cancer Energy Crisis → https://helping4cancer.com/cancer-energy-crisis/
How Radiation Therapy Kills Cancer Cells → https://helping4cancer.com/radiation-therapy-kills-cancer-cells/
Mitochondria and Apoptosis
One of the most important roles of mitochondria is controlling apoptosis, also known as programmed cell death.
How Apoptosis Works
In healthy cells, mitochondria release signals that trigger cell death when damage is detected. This process helps prevent the spread of abnormal cells.
Key components include:
- Cytochrome c release
- Activation of caspases
- Breakdown of cellular structures
Cancer Cells Block Apoptosis
Cancer cells often develop ways to avoid apoptosis, allowing them to survive longer than normal cells.
Common strategies include:
- Overexpression of anti-apoptotic proteins (like BCL-2)
- Suppression of pro-apoptotic signals
- Stabilization of mitochondrial membranes
By blocking apoptosis, cancer cells can continue growing even when damaged.
Related topic:
Apoptosis Resistance in Cancer → https://helping4cancer.com/apoptosis-resistance-cancer/
Mitochondrial Dysfunction in Tumors
Mitochondria in cancer cells are not completely broken—they are reprogrammed.
Structural and Functional Changes
Cancer-associated mitochondria may show:
- Altered membrane potential
- Changes in enzyme activity
- Mutations in mitochondrial DNA
These changes help tumors adapt to stress and hostile environments.
Hypoxia Adaptation
Tumors often grow in low-oxygen environments (hypoxia). Mitochondria help cancer cells survive by:
- Reducing oxygen consumption
- Shifting metabolic pathways
- Supporting anaerobic energy production
This adaptation is critical for tumor survival in poorly vascularized regions.
Mitochondria and Cancer Survival Pathways
Mitochondria interact with many key cancer survival pathways, including:
- PI3K/Akt (cell growth and survival)
- mTOR (nutrient sensing and metabolism)
- HIF-1α (response to low oxygen)
- NF-κB (inflammation and survival signaling)
These pathways coordinate energy production, stress responses, and immune evasion.
Related topic:
Why Cancer Cells Grow So Fast → https://helping4cancer.com/why-cancer-cells-grow-fast/
Targeting Mitochondria in Cancer Therapy
Because mitochondria are central to cancer survival, they are a promising target for treatment.
Disrupting Energy Production
Some therapies aim to:
- Inhibit mitochondrial respiration
- Reduce ATP production
- Starve cancer cells of energy
This can make tumors more vulnerable to treatment.
Increasing ROS
Another strategy is to push ROS levels beyond the threshold cancer cells can tolerate, leading to:
- Oxidative damage
- Mitochondrial collapse
- Cell death
Restoring Apoptosis
Therapies may also aim to reactivate apoptosis by:
- Targeting BCL-2 proteins
- Destabilizing mitochondrial membranes
- Triggering cytochrome c release
Combination Approaches
Modern strategies often combine mitochondrial targeting with:
- Chemotherapy
- Radiation therapy
- Metabolic therapies
These combinations can enhance treatment effectiveness.
The Future of Mitochondrial Research in Cancer
Research into mitochondria is rapidly evolving. Scientists are exploring:
- Mitochondrial-targeted drugs
- Personalized metabolic therapies
- Combination approaches that exploit metabolic weaknesses
Understanding mitochondrial behavior may lead to more precise and effective cancer treatments.
Key Takeaways
- Mitochondria are central to cancer cell energy, survival, and growth
- Cancer cells reprogram mitochondrial metabolism to adapt and thrive
- ROS produced by mitochondria can both support and destroy tumors
- Mitochria play a key role in controlling apoptosis, which cancer cells often evade
- Targeting mitochondrial function is a promising strategy in cancer therapy
External References
- National Cancer Institute – Cancer Metabolism
https://www.cancer.gov/research/key-initiatives/ras/ras-central/blog/2015/cancer-metabolism - Nature Reviews Cancer – Mitochondria in Cancer
https://www.nature.com/articles/nrc.2016.85 - Cell Metabolism – Targeting Mitochondria in Cancer
https://www.cell.com/cell-metabolism/home
Table of Contents