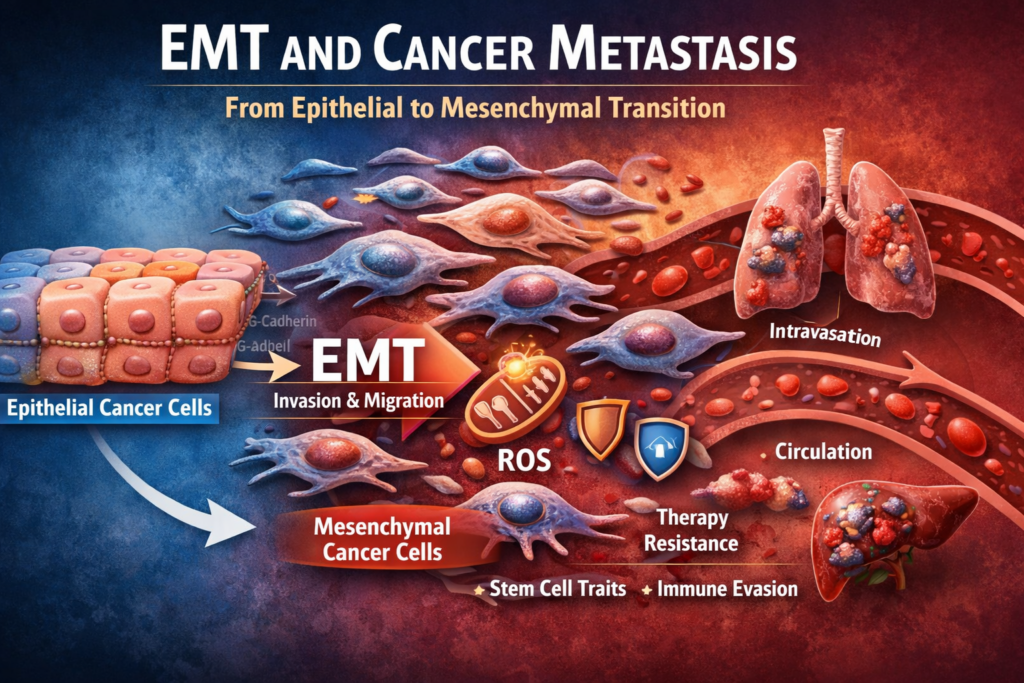
What Is EMT in Cancer?
Epithelial-to-mesenchymal transition (EMT) is a biological process where epithelial cells lose their structure and gain the ability to move, invade, and survive in new environments.
In normal biology, EMT plays a role in wound healing and embryonic development. In cancer, however, this same process is hijacked to enable metastasis.
Epithelial cells are typically:
- Tightly connected
- Structurally organized
- Non-mobile
During EMT, they transform into mesenchymal-like cells that are:
- Loosely connected
- Flexible and mobile
- Resistant to stress and therapy
This transition is one of the most important steps in cancer progression because it allows tumor cells to escape the primary tumor and spread throughout the body.
Why EMT Matters in Cancer Progression
Metastasis is responsible for the majority of cancer-related deaths. EMT is a key driver of this process.
When cancer cells undergo EMT, they gain several dangerous capabilities:
- Increased motility and invasion into surrounding tissue
- Ability to enter the bloodstream (intravasation)
- Resistance to cell death (apoptosis)
- Immune system evasion
- Stem-like properties that support tumor regrowth
EMT does not just help cancer spread—it also makes tumors harder to treat. Cells that undergo EMT are often more resistant to chemotherapy, radiation, and immune-based therapies.
This makes EMT a central target in understanding cancer survival strategies.
Key Molecular Changes During EMT
EMT involves a coordinated shift in gene expression, signaling pathways, and cellular behavior.
Loss of Epithelial Markers
Epithelial cells rely on proteins that maintain tight cell-to-cell adhesion.
Key changes include:
- Downregulation of E-cadherin
- Loss of tight junction proteins
- Breakdown of cell polarity
E-cadherin loss is one of the defining features of EMT and is strongly associated with increased tumor invasiveness.
Gain of Mesenchymal Markers
At the same time, cells begin expressing proteins associated with mobility and structural flexibility.
These include:
- N-cadherin
- Vimentin
- Fibronectin
This shift allows cancer cells to detach and move through surrounding tissues.
EMT Transcription Factors
Several transcription factors act as master regulators of EMT:
- Snail
- Slug
- Twist
- ZEB1 and ZEB2
These proteins suppress epithelial genes and activate mesenchymal programs, driving the full transition.
They are often activated by stress signals, inflammation, and tumor microenvironment factors.
EMT Signaling Pathways in Cancer
Multiple signaling pathways converge to drive EMT. These pathways are often already dysregulated in cancer.
TGF-beta Pathway
Transforming growth factor-beta (TGF-beta) is one of the most powerful EMT drivers.
It:
- Activates EMT transcription factors
- Promotes extracellular matrix remodeling
- Enhances invasion and migration
TGF-beta can act as a tumor suppressor early in cancer, but later becomes a promoter of metastasis.
PI3K/Akt Pathway
The PI3K/Akt pathway supports cell survival and metabolism.
In EMT, it:
- Enhances resistance to apoptosis
- Supports energy production for migration
- Interacts with other EMT pathways
This pathway is frequently activated in aggressive cancers.
Wnt/beta-catenin Pathway
Wnt signaling stabilizes beta-catenin, allowing it to enter the nucleus and activate EMT-related genes.
This pathway contributes to:
- Stem cell traits
- Tumor initiation
- Metastatic potential
NF-kB and Inflammation
Chronic inflammation strongly promotes EMT.
NF-kB signaling:
- Activates EMT transcription factors
- Promotes cytokine production
- Supports survival under stress
Inflammatory environments within tumors are a major driver of EMT activation.
EMT and Cancer Stem Cell Behavior
One of the most dangerous aspects of EMT is its link to cancer stem cells.
Cells undergoing EMT often gain stem-like properties, including:
- Self-renewal
- Ability to form new tumors
- Resistance to therapy
These cells are sometimes referred to as tumor-initiating cells.
They are thought to survive treatment and drive relapse.
EMT and stemness are tightly connected, meaning that targeting EMT may also reduce cancer recurrence.
Metabolic Changes During EMT
EMT is not just a structural transformation—it also involves deep metabolic reprogramming.
Shift Toward Glycolysis
Many EMT cells increase reliance on glycolysis, even in the presence of oxygen.
This provides:
- Rapid energy production
- Building blocks for growth
- Adaptation to hypoxic environments
This is often referred to as the Warburg effect.
Mitochondrial Adaptation
While glycolysis increases, mitochondria remain important.
During EMT:
- Mitochondria may become more flexible
- Reactive oxygen species (ROS) levels increase
- Cells adapt to oxidative stress
Moderate ROS levels can actually promote EMT by activating signaling pathways like TGF-beta and NF-kB.
ROS as a Driver of EMT
Reactive oxygen species play a dual role in cancer.
In EMT:
- Low to moderate ROS levels activate signaling pathways
- High ROS levels can kill cells
Cancer cells undergoing EMT often develop better antioxidant defenses to survive oxidative stress.
This balance allows them to use ROS as a signaling tool without triggering cell death.
EMT and the Tumor Microenvironment
The tumor microenvironment plays a major role in triggering EMT.
Key contributors include:
- Hypoxia (low oxygen levels)
- Inflammatory cytokines
- Cancer-associated fibroblasts
- Immune cells
Hypoxia activates HIF-1alpha, which promotes EMT and metabolic adaptation.
Fibroblasts remodel the extracellular matrix, making it easier for cancer cells to invade.
Immune cells release signals that can either suppress or promote EMT depending on context.
This environment acts as a training ground for metastatic cancer cells.
EMT and Immune Evasion
Cancer cells undergoing EMT become less visible to the immune system.
They may:
- Reduce antigen presentation
- Increase PD-L1 expression
- Suppress immune cell activity
This allows them to escape immune surveillance and survive in circulation.
EMT is therefore closely linked to immune resistance and failure of immunotherapy in some patients.
EMT and Treatment Resistance
EMT contributes to resistance across multiple treatment types.
Chemotherapy Resistance
EMT cells:
- Divide more slowly
- Activate survival pathways
- Increase drug efflux
This makes them harder to kill with standard chemotherapy.
Radiation Resistance
Radiation relies partly on ROS to damage cancer cells.
EMT cells often:
- Improve antioxidant defenses
- Repair DNA damage more effectively
This reduces the effectiveness of radiation therapy.
Targeted Therapy Resistance
EMT can bypass targeted therapies by:
- Activating alternative pathways
- Changing receptor expression
- Adapting metabolism
This plasticity makes EMT-driven cancers highly adaptable.
Partial EMT and Cellular Plasticity
EMT is not always a complete transformation.
Many cancer cells exist in a hybrid state known as partial EMT.
These cells:
- Retain some epithelial features
- Gain some mesenchymal traits
- Are highly adaptable
Partial EMT may be even more dangerous because it allows cells to:
- Travel efficiently
- Revert back (MET) to form new tumors
This dynamic flexibility is a major challenge in cancer treatment.
EMT and Metastatic Colonization
After traveling through the bloodstream, cancer cells often reverse EMT through a process called mesenchymal-to-epithelial transition (MET).
This allows them to:
- Reattach to tissue
- Proliferate
- Form secondary tumors
EMT enables escape, while MET enables colonization.
This reversible process highlights the adaptability of cancer cells.
Clinical Importance of Targeting EMT
Because EMT is central to metastasis and resistance, it is a major focus of cancer research.
Potential strategies include:
- Inhibiting TGF-beta signaling
- Targeting EMT transcription factors
- Modulating tumor metabolism
- Reducing inflammation
However, EMT is complex and tightly integrated into normal biology, making it difficult to target without side effects.
Combination approaches may be necessary.
Key Takeaways
- EMT enables cancer cells to become mobile, invasive, and resistant
- It is driven by pathways like TGF-beta, PI3K/Akt, and NF-kB
- EMT is closely linked to cancer stem cells and relapse
- Metabolic changes, including ROS signaling and mitochondrial adaptation, support EMT
- The tumor microenvironment plays a major role in activating EMT
- EMT contributes to immune evasion and treatment resistance
- Partial EMT and cellular plasticity make cancer more adaptable
Understanding EMT is essential for understanding how cancer spreads and survives treatment.
External References
National Cancer Institute – https://www.cancer.gov
NIH EMT Overview – https://www.ncbi.nlm.nih.gov
Nature Reviews Cancer EMT Article – https://www.nature.com
PubMed EMT and Metastasis Research – https://pubmed.ncbi.nlm.nih.gov
Cancer Research Journal – https://aacrjournals.org
Internal Links
https://helping4cancer.com/tumor-survival-network/
https://helping4cancer.com/cancer-and-mitochondria/
https://helping4cancer.com/cancer-oxidative-stress/
https://helping4cancer.com/cancer-cell-cycle/
https://helping4cancer.com/tumor-immune-cloaking/