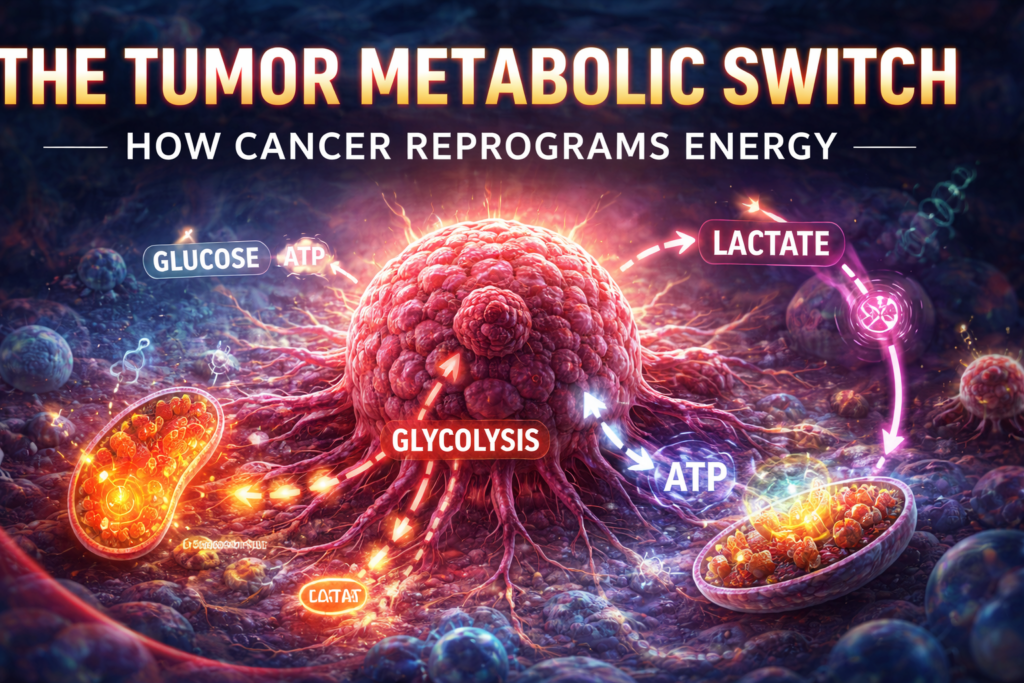
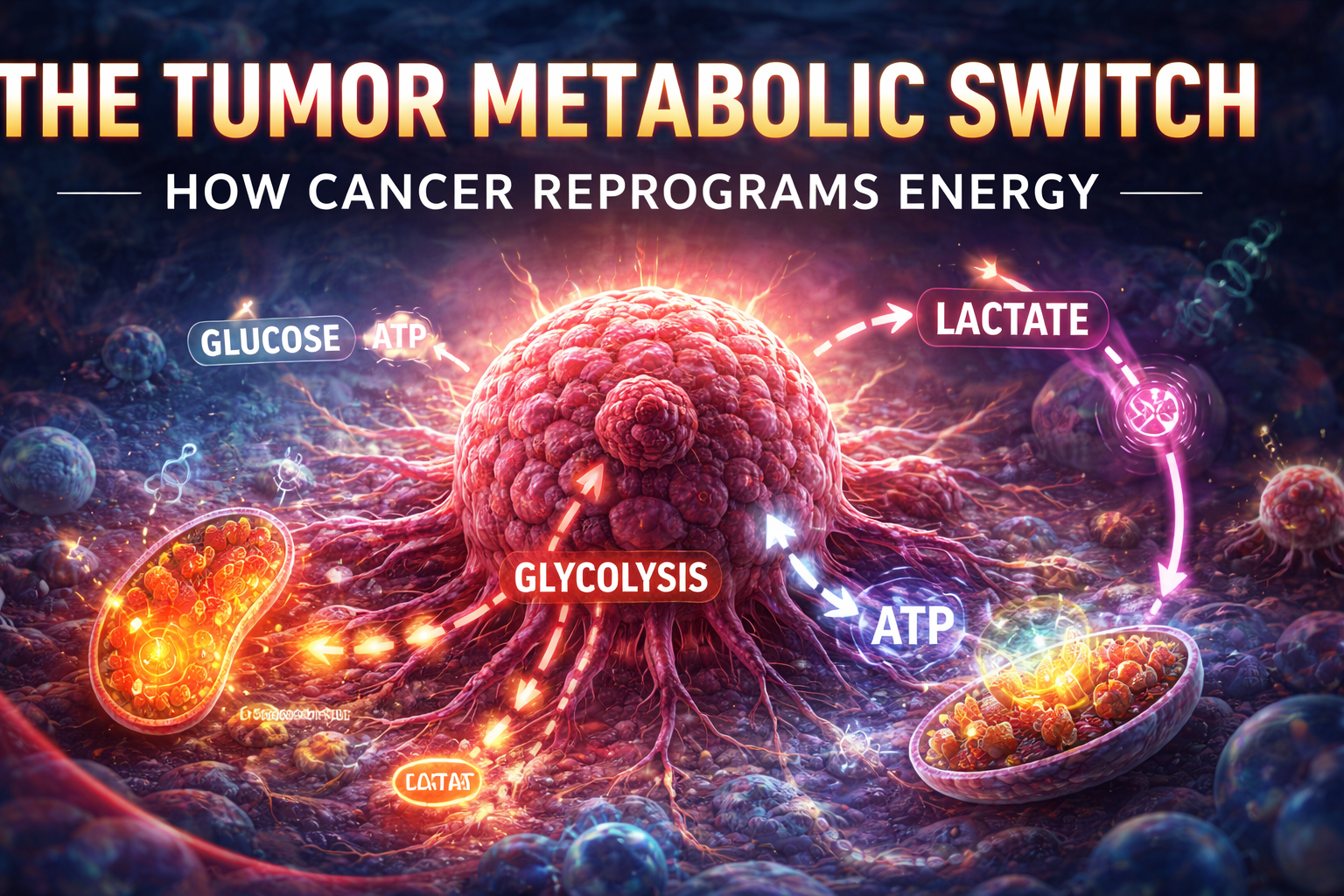
Cancer is not only a disease of uncontrolled cell growth—it is also a disease of altered energy metabolism. One of the defining features of cancer cells is their ability to reprogram how they generate and use energy, a process known as the tumor metabolic switch.
This metabolic shift allows cancer cells to grow rapidly, survive under stress, and adapt to hostile environments inside the body. Unlike normal cells, which rely heavily on efficient mitochondrial energy production, cancer cells often switch to alternative pathways such as glycolysis and flexible fuel usage.
Understanding the tumor metabolic switch is essential because many modern therapies are now designed to target cancer metabolism and exploit its weaknesses.
What Is the Tumor Metabolic Switch?
The tumor metabolic switch refers to the ability of cancer cells to alter their energy production systems to support continuous growth and survival.
Healthy cells primarily generate energy through oxidative phosphorylation, a highly efficient process that takes place in the mitochondria. This system produces large amounts of ATP using oxygen.
Cancer cells, however, often shift toward aerobic glycolysis, meaning they generate energy through glycolysis even when oxygen is available.
This phenomenon is known as the Warburg Effect and is a hallmark of cancer metabolism.
👉 Internal Link:
https://helping4cancer.com/cancer-cells-love-sugar/
While glycolysis produces less ATP per molecule of glucose, it allows cancer cells to rapidly produce the building blocks needed for growth, including nucleotides, amino acids, and lipids.
Why Cancer Cells Reprogram Their Metabolism
Cancer cells do not randomly switch their metabolism—this process provides several key survival advantages.
Rapid Growth and Proliferation
Tumor cells divide quickly and require massive amounts of raw materials to build new cells.
Metabolic reprogramming allows cancer cells to produce:
- DNA for replication
- proteins for structure and function
- lipids for cell membranes
By favoring glycolysis and related pathways, cancer cells can generate these materials faster than normal cells.
Survival in Low Oxygen (Hypoxia)
As tumors grow, they often outpace their blood supply, creating low oxygen environments.
Under these conditions, mitochondrial respiration becomes less effective.
Glycolysis, however, does not require oxygen, making it an ideal energy source for tumors in hypoxic conditions.
This adaptation is driven in part by hypoxia-related signaling pathways.
👉 Internal Link:
https://helping4cancer.com/tumor-microenvironment/
Resistance to Cell Death
Mitochondria play a central role in apoptosis, or programmed cell death.
By reducing reliance on mitochondrial respiration, cancer cells can limit apoptosis signaling, helping them survive longer and resist treatments.
👉 Internal Link:
https://helping4cancer.com/apoptosis-resistance-cancer/
Glycolysis: The Core of the Tumor Metabolic Switch
Glycolysis is one of the most important metabolic pathways in cancer.
In normal cells, glycolysis is a temporary backup system used when oxygen is scarce. In cancer cells, it often becomes the primary energy pathway.
Key Features of Glycolysis in Cancer
- Increased glucose uptake
- Overexpression of glycolytic enzymes
- Conversion of glucose to lactate
- Reduced reliance on mitochondrial respiration
This is why cancer cells consume large amounts of sugar, which can be visualized using PET scans.
👉 External Reference:
https://www.cancer.gov/about-cancer/understanding/what-is-cancer
The Warburg Effect Explained
The Warburg Effect describes how cancer cells preferentially use glycolysis over oxidative phosphorylation, even when oxygen is present.
This metabolic shift allows cancer cells to:
- produce energy quickly
- generate biosynthetic precursors
- support rapid tumor expansion
Although inefficient in terms of ATP yield, glycolysis provides a growth advantage.
Mitochondrial Dysfunction in Cancer
While glycolysis plays a central role, mitochondria are not inactive in cancer cells. Instead, they become reprogrammed.
How Mitochondria Change in Cancer
Cancer-associated mitochondrial changes include:
- altered electron transport chain activity
- increased reactive oxygen species (ROS) production
- mutations in mitochondrial DNA
- shifts in metabolic signaling pathways
Rather than serving only as energy generators, mitochondria become biosynthetic and signaling hubs.
👉 Internal Link:
https://helping4cancer.com/reactive-oxygen-species-cancer/
Mitochondria and Oxidative Stress
Cancer cells often operate under elevated levels of oxidative stress.
Reactive oxygen species (ROS) can promote:
- DNA mutations
- tumor progression
- metabolic adaptation
However, excessive ROS can also damage cancer cells, which is why some therapies aim to push ROS levels beyond survivable thresholds.
Metabolic Flexibility: Cancer’s Survival Advantage
One of the most dangerous aspects of cancer metabolism is metabolic flexibility.
Cancer cells can switch between different fuel sources depending on what is available.
Alternative Energy Sources Used by Cancer Cells
Glucose
The primary fuel for glycolysis.
Glutamine
Supports the TCA cycle and biosynthesis.
👉 Internal Link:
https://helping4cancer.com/why-cancer-cells-need-glutamine/
Fatty Acids
Used for membrane production and energy storage.
Iron-Dependent Metabolism
Supports mitochondrial function and DNA synthesis.
👉 Internal Link:
https://helping4cancer.com/cancer-cells-need-iron/
Why Metabolic Flexibility Matters
This adaptability allows tumors to:
- survive nutrient deprivation
- resist therapies
- grow in diverse environments
Metabolic flexibility is one of the key reasons cancer is difficult to treat.
Lactate and the Tumor Microenvironment
A major byproduct of glycolysis is lactate, which plays an important role in tumor biology.
Effects of Lactate in Cancer
Lactate can:
- acidify the tumor environment
- suppress immune cell function
- promote angiogenesis
- enhance tumor invasion
Instead of being waste, lactate acts as a signaling molecule that supports tumor survival.
👉 External Reference:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6520897/
The Tumor Microenvironment and Metabolism
The tumor metabolic switch is influenced by the surrounding environment.
The tumor microenvironment includes:
- immune cells
- blood vessels
- fibroblasts
- signaling molecules
These components affect how cancer cells access nutrients and respond to stress.
👉 Internal Link:
https://helping4cancer.com/tumor-microenvironment/
Targeting the Tumor Metabolic Switch
Because cancer cells depend on altered metabolism, researchers are exploring therapies that target these pathways.
Metabolic Therapy Strategies
Targeting Glycolysis
Blocking glycolytic enzymes may reduce tumor energy production.
Nutrient Restriction
Approaches such as fasting may influence cancer metabolism.
👉 Internal Link:
https://helping4cancer.com/fasting-cancer-therapy/
Increasing Oxidative Stress
Some therapies aim to increase ROS to toxic levels in cancer cells.
👉 Internal Link:
https://helping4cancer.com/cancer-metabolic-trap/
Why Metabolic Targeting Works
Cancer cells operate close to their metabolic limits. By disrupting their energy systems, therapies may:
- weaken tumor survival
- increase treatment sensitivity
- promote cancer cell death
Why the Tumor Metabolic Switch Matters
The tumor metabolic switch is one of the most important concepts in modern cancer research.
It explains how cancer cells:
- grow rapidly
- survive under stress
- adapt to different environments
- resist therapy
By targeting metabolic pathways, scientists aim to cut off the energy supply that tumors depend on.
Key Takeaways
- Cancer cells reprogram energy production
- Glycolysis often replaces mitochondrial respiration
- Mitochondria become signaling hubs
- Tumors develop metabolic flexibility
- Lactate supports tumor growth and immune evasion
- Metabolic therapies aim to exploit these weaknesses
Understanding the tumor metabolic switch provides insight into how cancer survives—and how it may be targeted more effectively.
External References
National Cancer Institute
https://www.cancer.gov
Nature Reviews Cancer – Metabolic Reprogramming
https://www.nature.com/articles/nrc.2016.60
NIH – Cancer Metabolism Overview
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6520897/
Table of Contents