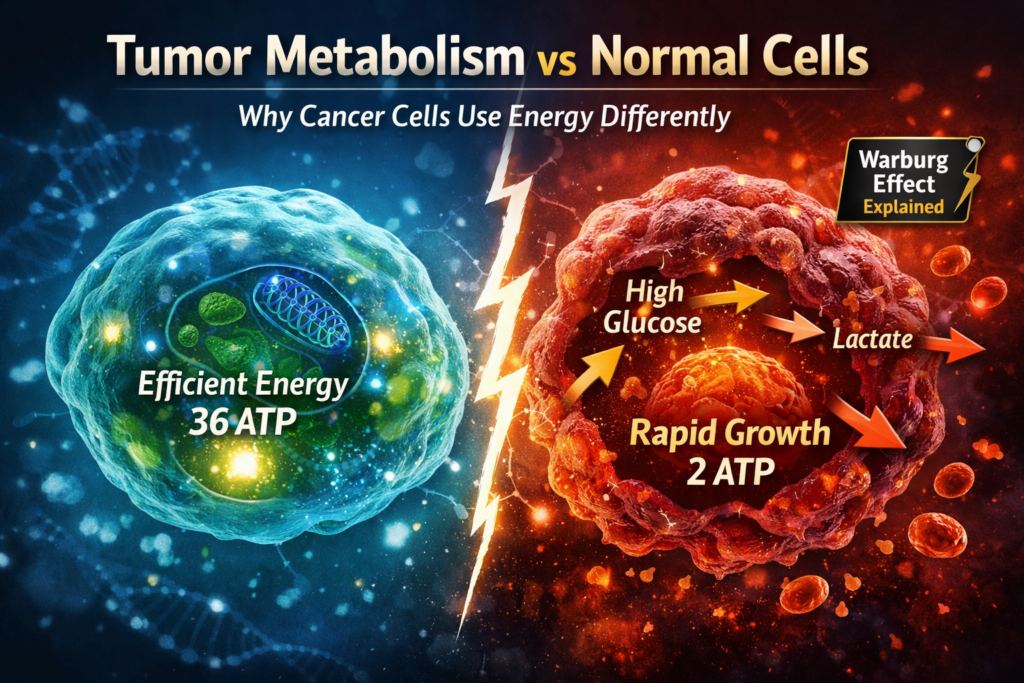
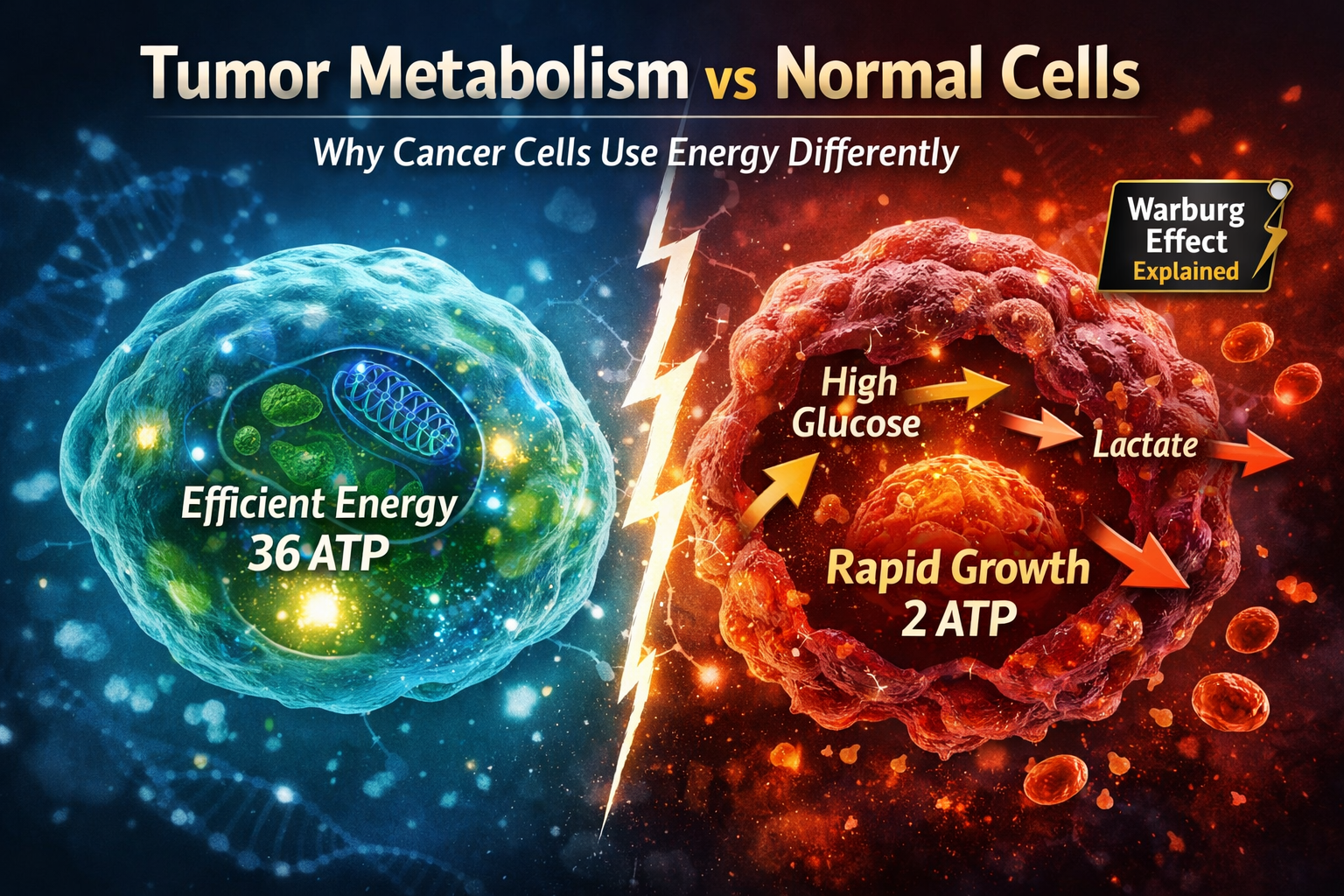
Cancer is not just a disease of uncontrolled growth—it is also a disease of altered metabolism. One of the most important differences between healthy cells and cancer cells lies in how they produce and use energy.
Normal cells rely on highly efficient metabolic systems to generate energy. In contrast, tumor cells reprogram their metabolism to support rapid growth, survival under stress, and resistance to treatment.
Understanding tumor metabolism compared to normal metabolism reveals key vulnerabilities that modern therapies—and metabolic strategies—aim to target.
What Normal Cell Metabolism Looks Like
Efficient Energy Production
Healthy cells generate energy primarily through oxidative phosphorylation inside the mitochondria.
In this process:
- Glucose is broken down into pyruvate
- Pyruvate enters the mitochondria
- Oxygen is used to produce ATP
This pathway is highly efficient, producing up to 36 ATP molecules per glucose molecule.
Metabolic Flexibility
Normal cells can switch between different fuel sources depending on availability:
- Glucose
- Fatty acids
- Ketones
This flexibility allows cells to adapt to fasting, exercise, and metabolic stress without damage.
What Tumor Metabolism Looks Like
The Warburg Effect
A defining feature of cancer metabolism is the Warburg effect, where cancer cells rely heavily on glycolysis even when oxygen is available.
Instead of fully using mitochondria, cancer cells:
- Convert glucose into lactate
- Produce only about 2 ATP per glucose
- Consume large amounts of glucose
External reference:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4783224/
Why Tumors Prefer Glycolysis
Although inefficient, glycolysis supports cancer growth because it:
- Produces energy quickly
- Generates building blocks for cell division
- Helps survival in low-oxygen environments
This metabolic shift is one of the core hallmarks of cancer.
Key Differences Between Tumor and Normal Metabolism
Glucose Consumption
Tumor cells consume significantly more glucose than normal cells.
- Tumors: High glucose uptake (visible on PET scans)
- Normal cells: Controlled glucose use
Internal link:
/glycolysis-cancer/
Mitochondrial Function
Normal cells rely heavily on mitochondria for energy production. Cancer cells often show mitochondrial dysfunction.
In tumor cells:
- Mitochondria are altered or partially impaired
- Oxidative phosphorylation is reduced
- Reactive oxygen species (ROS) are elevated
Mitochondria are not inactive—they are repurposed to support growth and survival.
Energy Efficiency vs Speed
Normal cells prioritize efficiency. Cancer cells prioritize speed.
- Normal cells: High ATP yield
- Tumor cells: Rapid but inefficient energy production
This trade-off supports fast tumor growth.
Metabolic Flexibility vs Dependency
Normal cells can adapt to multiple fuels. Tumor cells are often dependent on:
- Glucose
- Glutamine
This dependency creates therapeutic opportunities.
Internal link:
/cancer-cells-need-glutamine/
Lactate Production
Cancer cells produce large amounts of lactate even when oxygen is present.
This leads to:
- Acidic tumor environments
- Immune suppression
- Increased invasion and metastasis
Internal link:
/tumor-microenvironment/
Mitochondrial Dysfunction in Cancer
Altered Role of Mitochondria
In cancer cells, mitochondria shift away from efficient energy production and instead support:
- Biosynthesis
- Redox balance
- Survival signaling
Reactive Oxygen Species (ROS)
Tumor cells often operate with elevated ROS levels.
- Moderate ROS supports tumor growth
- High ROS can trigger cell death
This creates a fragile balance that therapies aim to exploit.
Internal link:
/reactive-oxygen-species-cancer/
Metabolic Stress in Tumor Cells
What Is Metabolic Stress
Metabolic stress occurs when cells struggle to maintain energy production or manage oxidative damage.
Tumor cells constantly face stress due to:
- Rapid growth
- Poor blood supply
- Low oxygen (hypoxia)
- Nutrient competition
Tumor Adaptation
Cancer cells adapt by:
- Increasing glucose uptake
- Activating survival pathways such as PI3K/Akt and AMPK
- Rewiring metabolic processes
These adaptations help survival but also create vulnerabilities.
Why Tumor Metabolism Matters for Treatment
Targeting Glucose Metabolism
Some therapies aim to disrupt glycolysis and reduce glucose availability to tumors.
Exploiting ROS Vulnerability
Cancer cells operate near oxidative limits. Increasing ROS can push them into cell death.
Metabolic Therapy Approaches
Emerging strategies include:
- Fasting and metabolic stress
- Ketogenic approaches
- Targeting metabolic pathways
Internal links:
/fasting-cancer-therapy/
/ketones-vs-cancer/
/cancer-metabolic-trap/
Summary Comparison
| Feature | Normal Cells | Tumor Cells |
|---|---|---|
| Energy Source | Flexible | Glucose-dependent |
| ATP Production | Efficient (36 ATP) | Inefficient (2 ATP) |
| Mitochondria | Fully functional | Altered |
| Lactate | Low | High |
| ROS Levels | Controlled | Elevated |
| Growth | Regulated | Rapid |
Final Thoughts
The differences between tumor metabolism and normal metabolism form the foundation of many modern cancer research strategies.
Cancer cells sacrifice efficiency for speed, flexibility for dependency, and stability for growth. These trade-offs create weaknesses that may be targeted through both conventional treatments and metabolic approaches.
Understanding these differences provides a clearer picture of how cancer survives—and where it may be most vulnerable.
External References
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4783224/
https://www.cancer.gov/about-cancer/understanding/what-is-cancer
https://www.nature.com/articles/nrc.2017.10
Table of Contents