Introduction: What Fenbendazole Is and Why It Matters in Cancer
Fenbendazole is a benzimidazole antiparasitic drug commonly used in veterinary medicine. It was developed for deworming animals, not for cancer treatment. Interest in fenbendazole and cancer grew after anecdotal reports suggested possible benefit, but the current evidence is still mostly preclinical. There are no large human clinical trials proving fenbendazole treats cancer.
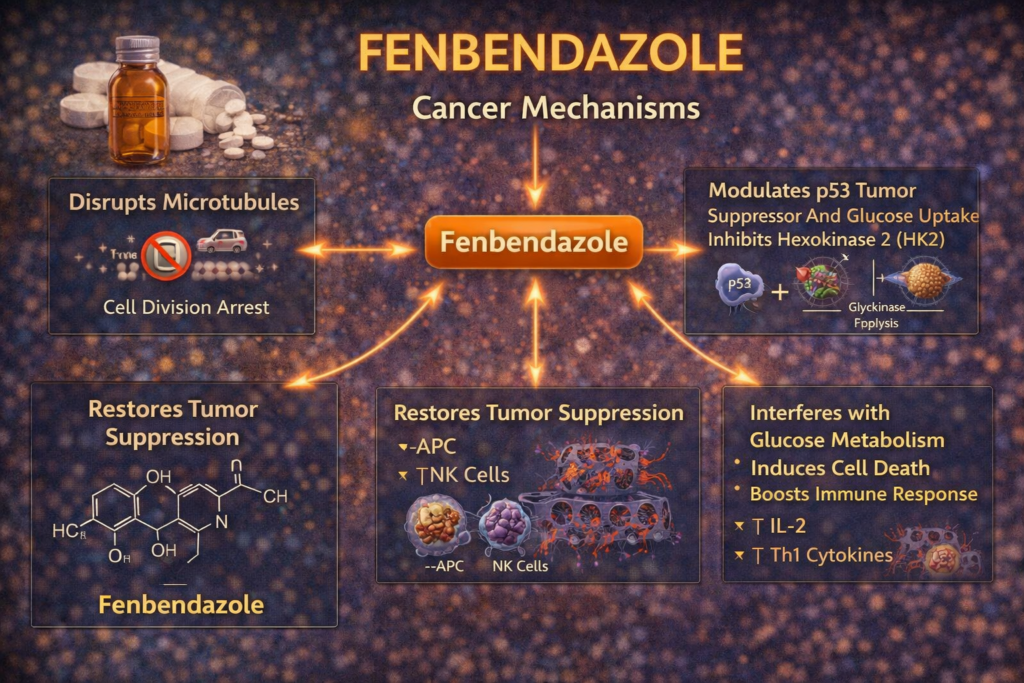
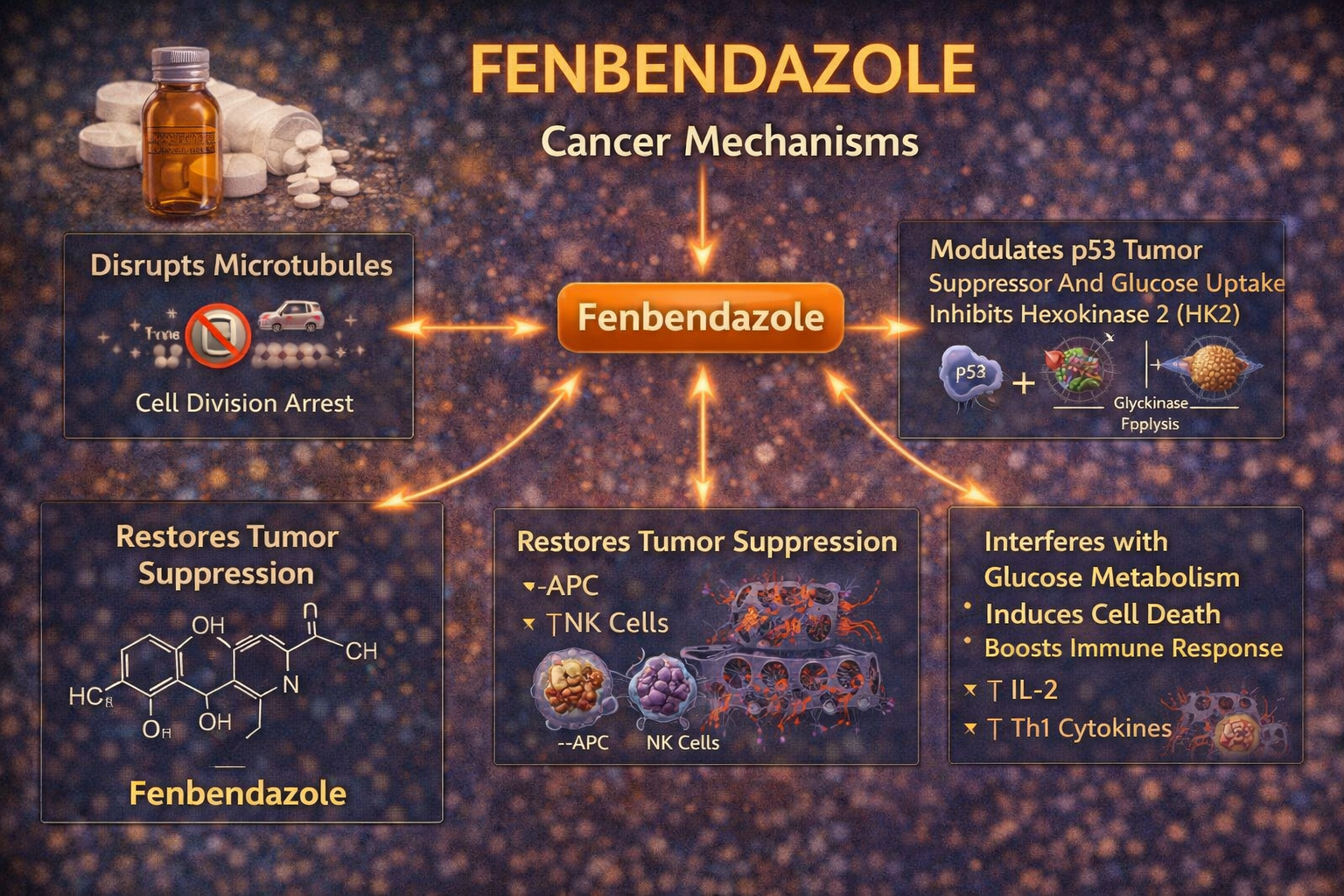
What makes fenbendazole interesting in cancer research is that it appears to affect several tumor survival systems at once. Experimental studies suggest it can disrupt microtubules, reduce glucose uptake, increase oxidative stress in cancer cells, and in some settings promote apoptosis or ferroptosis. That places it within a broader metabolic and pathway-based cancer discussion rather than as a proven therapy.
To understand where this fits into the bigger system, start here:
The foundation of cancer
https://helping4cancer.com/the-foundation-of-cancer/
What Fenbendazole Is
Fenbendazole belongs to the same general benzimidazole family as mebendazole and albendazole. Its best-established use is antiparasitic treatment, and its safety profile is based mainly on animal use, not cancer treatment in humans. A 2024 review notes growing interest in repurposing it for oncology, but also emphasizes the lack of human efficacy data and the need for better pharmacokinetic and safety understanding.
That distinction matters. Fenbendazole may be biologically active against cancer in the lab, but that is very different from saying it is clinically proven in people.
How Fenbendazole Works in Cancer
Pathways: Microtubules, Cell Cycle, and Survival Signaling
One of fenbendazole’s best-documented actions is microtubule disruption. Microtubules act like the internal framework and transport system of a cell. Cancer cells depend on them for division, movement, and survival. In a 2018 study, fenbendazole showed moderate tubulin-binding activity and disrupted microtubule dynamics in human cancer cells.
That microtubule effect helps explain several downstream results:
- cell cycle arrest, especially around the G2/M checkpoint
- mitotic stress
- apoptosis signaling
- reduced tumor proliferation
Experimental work also suggests fenbendazole may influence PI3K/Akt/mTOR and MAPK-related survival signaling indirectly through broader cellular stress and metabolic disruption. Your original page is directionally consistent here, but the strongest direct evidence is still around microtubules, glucose metabolism, oxidative stress, and cell cycle effects.
For related pathway context, see:
PI3K/Akt pathway and tumor survival
https://helping4cancer.com/pi3k-akt-pathway-cancer/
Metabolism: Glucose, Glycolysis, and Tumor Fuel
Fenbendazole is especially relevant from a metabolic perspective because studies suggest it can interfere with glucose uptake and glycolysis. The 2018 Scientific Reports study found reduced glucose uptake, lower GLUT transporter expression, and downregulation of hexokinase II, a major enzyme that many cancer cells rely on for rapid energy production.
That matters because many tumors depend on the Warburg effect, using glucose aggressively even when oxygen is available. By interfering with sugar uptake and glycolytic machinery, fenbendazole may add metabolic stress to cancer cells, especially in low-glucose or fasting conditions.
This is why fenbendazole fits naturally into discussions about:
Cancer metabolism
https://helping4cancer.com/cancer-metabolism/
Fasting and metabolic therapy
https://helping4cancer.com/fasting-cancer-plan/
https://helping4cancer.com/metabolic-therapy-cancer/
Immune System: Indirect Support Through Tumor Weakening
Fenbendazole is not primarily known as an immune stimulant like a beta-glucan mushroom or an NK-cell-focused herb. Its immune relevance is more indirect. By weakening tumor metabolism, increasing oxidative stress, and interfering with survival signaling, it may make tumors less able to resist immune pressure. Experimental reviews also discuss broader biologic effects on tumor burden and vascularity, but direct human immune-support evidence is lacking.
So within a larger cancer system, fenbendazole is better understood as an attack-phase stressor than a recovery-phase immune support compound.
For broader immune context, see:
Immune system and cancer defense
https://helping4cancer.com/immune-system-cancer/
The Three Main Anti-Cancer Mechanisms
1. Microtubule Disruption
Fenbendazole interferes with tubulin and destabilizes microtubules, making it harder for cancer cells to divide and survive. This is one of the strongest experimentally supported mechanisms.
2. Glucose Metabolism Inhibition
Fenbendazole reduces glucose uptake and suppresses glycolysis-related machinery such as GLUT transporters and HKII. This weakens the tumor’s fuel supply and increases vulnerability under metabolic stress.
3. Glutathione and Oxidative Stress Pressure
Your original page emphasizes glutathione depletion. The 2024 review supports fenbendazole’s role in inducing oxidative stress and discusses its interaction with glycolysis and apoptosis, while broader cancer literature shows glutathione is central to treatment resistance. The strongest direct fenbendazole evidence here is that it increases oxidative stress and can contribute to ferroptosis-related death in resistant colorectal cancer models.
That makes fenbendazole relevant to:
Redox balance and oxidative stress
https://helping4cancer.com/redox-balance-cancer/
https://helping4cancer.com/oxidative-stress-cancer/
Ferroptosis, ROS, and Cell Death
Fenbendazole has also been linked to ferroptosis in 5-FU-resistant colorectal cancer cells. The 2022 study found increased ferroptosis and ferroptosis-augmented apoptosis in resistant cells, suggesting fenbendazole may help exploit a vulnerability that some resistant tumors still have.
This matters because many tumors become resistant to standard apoptosis pathways. A compound that adds oxidative stress and pushes cells toward ferroptosis may help widen the pressure on treatment-resistant disease.
Role in Cancer Strategy
Fenbendazole fits best into an attack-phase or metabolic-stress role, not a recovery-phase antioxidant role.
Where It Fits Best
Fenbendazole makes the most sense in strategies centered on:
- metabolic ignition
- fasting-related tumor stress
- glycolysis disruption
- oxidative pressure
- attack-phase stacking with other tumor-stressing agents
That is because its proposed value comes from weakening cancer cells when glucose is low and oxidative pressure is high.
Connection to Fasting and Oxidative Stress
My original page places fenbendazole in a fasted early-morning window. The reasoning is consistent with its preclinical biology: lower glucose availability, more metabolic stress, and potentially greater vulnerability to oxidative damage. That said, this timing framework is theoretical and protocol-based, not something validated in human cancer trials.
So the most accurate framing is:
- it is biologically plausible in a fasting-plus-attack model
- it is not clinically proven as a timed human cancer therapy
Protocol 2 Placement
Within the Protocol 2 framework I provided, fenbendazole is used during the Metabolic Ignition phase, usually in the fasted state and with fat for better absorption.
Why Fats Matter
Fenbendazole has poor water solubility. Review literature and experimental delivery research support the idea that bioavailability is a major limitation, which is one reason lipid-based systems and nanoparticles are being studied.
That means taking fenbendazole with fat is a practical strategy within the protocol logic, even though optimized over-the-counter human formulations are still not standardized.
Suggested Protocol Logic From the Existing Page
My existing page places fenbendazole roughly like this:
- 500 to 1000 mg at 6:30 AM with fat
- optional second 500 mg dose later in the morning
The rationale is:
- fasted state
- lower glucose availability
- stronger metabolic stress
- alignment with an oxidative attack window
That framework preserves my original meaning, but it should be presented as a protocol-specific strategy, not a medically established dosing standard.
Interaction With Radiation
My original page presents fenbendazole as a radiosensitizer. The evidence here is mixed. Some experimental work and reviews discuss oxidative stress and tumor weakening, but a mouse EMT6 model found no radiosensitizing effect from fenbendazole-containing diets.
So the most accurate statement is:
Fenbendazole may theoretically enhance radiation response through oxidative and metabolic mechanisms, but existing experimental data are conflicting, and this has not been established clinically.
Fenbendazole Compared With Mebendazole and Albendazole
Fenbendazole, mebendazole, and albendazole all belong to the benzimidazole family, but they are not interchangeable in every context.
A careful comparison is:
- Fenbendazole: strong preclinical interest, low cost, veterinary availability, but no established human oncology role
- Mebendazole: more human clinical familiarity overall and more attention in repurposing discussions, especially in brain tumor literature
- Albendazole: used in humans for parasitic disease, similar family mechanism, but less commonly discussed in patient-led cancer repurposing circles
Your original page is directionally reasonable here, but this comparison should stay cautious unless tied to specific clinical contexts.
Safety, Risks, and Supervision
Fenbendazole is often described as having a wide safety margin in animals, but that should not be confused with proven safety for chronic off-label human cancer use. The 2024 review discusses toxicity considerations and the need for careful evaluation, and case-based literature stresses physician oversight.
Important safety points include:
- possible liver injury
- possible gastrointestinal upset
- uncertain drug interactions in complex cancer regimens
- variable product quality outside regulated pharmaceutical pathways
This is why any off-label use should involve clinician input and liver monitoring.
Key Benefits Being Studied
- microtubule disruption
- cell cycle arrest
- apoptosis support
- glycolysis and glucose uptake inhibition
- oxidative stress increase in tumor cells
- possible ferroptosis support in resistant colorectal cancer
- possible synergy with fasting-based metabolic pressure
- possible reduction in tumor size or vascularity in some animal models
All of these are research signals, not confirmed clinical benefits.
Realistic Expectations
The current evidence does not support fenbendazole as a proven cancer cure. The strongest data are from cell lines, animal models, reviews of repurposing potential, and anecdotal or self-use reports. Human evidence is still inadequate.
So the most honest conclusion is:
Fenbendazole is a promising experimental repurposing candidate with plausible anti-cancer mechanisms, but it remains unproven in large human trials.
Final Thoughts
Fenbendazole is being discussed in cancer because it appears to hit several important tumor vulnerabilities at once:
- microtubules
- glucose metabolism
- oxidative stress
- ferroptosis-related death
- cell cycle control
That makes it a strong fit conceptually for a larger metabolic and oxidative cancer strategy. But concept and proof are not the same. Fenbendazole still lacks the human trial evidence needed to treat it as established cancer therapy. Used carefully, it may be viewed as a protocol-specific support tool under medical supervision, not a replacement for standard care.
Related Topics
- The foundation of cancer
https://helping4cancer.com/the-foundation-of-cancer/ - Cancer metabolism
https://helping4cancer.com/cancer-metabolism/ - PI3K/Akt pathway and tumor survival
https://helping4cancer.com/pi3k-akt-pathway-cancer/ - Redox balance and oxidative stress
https://helping4cancer.com/redox-balance-cancer/
https://helping4cancer.com/oxidative-stress-cancer/ - Fasting and metabolic cancer strategy
https://helping4cancer.com/fasting-cancer-plan/
https://helping4cancer.com/metabolic-therapy-cancer/ - EMT and metastasis
https://helping4cancer.com/emt-cancer-metastasis/
Research References
🧬 Microtubule Disruption & Antiproliferative Effects
- Fenbendazole as a moderate microtubule destabilizing agent… blocks growth of human xenografts in mice pmc.ncbi.nlm.nih.gov+15pubmed.ncbi.nlm.nih.gov+15ar.iiarjournals.org+15
- Sci Rep 2018: Fenbendazole causes cancer cell death by modulating multiple pathways en.wikipedia.org+2pmc.ncbi.nlm.nih.gov+2pmc.ncbi.nlm.nih.gov+2
🍭 Glucose Metabolism Inhibition
- Same study highlights: inhibited glucose uptake, GLUT transporter expression, HKII downregulation pubmed.ncbi.nlm.nih.gov+3pmc.ncbi.nlm.nih.gov+3pubmed.ncbi.nlm.nih.gov+3
🔰 Ferroptosis & Oxidative Stress
- Fenbendazole induces ferroptosis in 5-FU–resistant colorectal cancer cells, boosting iron-dependent cell death pmc.ncbi.nlm.nih.gov+15pubmed.ncbi.nlm.nih.gov+15pmc.ncbi.nlm.nih.gov+15
- Ferroptosis: a non-apoptotic, iron-dependent form of cell death with tumor suppression potential pmc.ncbi.nlm.nih.gov+15pubmed.ncbi.nlm.nih.gov+15pubmed.ncbi.nlm.nih.gov+15
🔁 Cell Cycle Arrest & Apoptosis
- Fenbendazole suppresses growth and induces apoptosis via p21-mediated cell cycle arrest in liver cancer cells (H4IIE) pmc.ncbi.nlm.nih.gov+4pubmed.ncbi.nlm.nih.gov+4pubmed.ncbi.nlm.nih.gov+4
📉 Broader Anticancer Review & Bioavailability
- “Oral Fenbendazole for Cancer Therapy in Humans and Animals” (2024) – covers PK, glycolysis, apoptosis, oxidative stress, and safety pubmed.ncbi.nlm.nih.gov+1ar.iiarjournals.org+1
- “An Update on the Biologic Effects of Fenbendazole” – shows tumor size/vascularity decrease and confirms microtubule targeting pmc.ncbi.nlm.nih.gov
⚠️ Conflicting Radiation Data
- Fenbendazole showed no radiosensitizing effect in EMT6 mouse tumor model pmc.ncbi.nlm.nih.gov+15pmc.ncbi.nlm.nih.gov+15pubmed.ncbi.nlm.nih.gov+15
🐕 Comparative & Canine Data
- G2/M arrest and mitotic slippage in canine melanoma cell lines en.wikipedia.org+7pubmed.ncbi.nlm.nih.gov+7pubmed.ncbi.nlm.nih.gov+7
📌 Epidemiology & User Cases
- “Exceptional Repositioning of Dog Dewormer: Fenbendazole Fever” – notes widespread self-use and the importance of physician oversight pubmed.ncbi.nlm.nih.gov+1pubmed.ncbi.nlm.nih.gov+1
Table of Contents