Introduction: What Gingerol Is and Why It Matters in Cancer
Gingerol, especially 6-gingerol, is the main pungent phenolic compound in fresh ginger root. It gives ginger its spicy kick, but it also has strong biological activity that has made it a major focus in cancer research. Preclinical studies suggest gingerol can influence apoptosis, inflammation, oxidative stress, metastasis, angiogenesis, and several tumor survival pathways. Research has been especially active in digestive cancers such as colorectal, gastric, pancreatic, and liver cancer.
What makes gingerol especially interesting is that it does not act through only one mechanism. It appears to work across inflammation, metabolism, and cell-death signaling at the same time. That multi-target profile is why it fits naturally into a larger cancer system rather than as a stand-alone compound. For the bigger picture, see:
https://helping4cancer.com/the-foundation-of-cancer/
What Gingerol Is
Gingerol is one of the major active constituents of fresh ginger, while shogaols become more prominent in dried or heated ginger. Among these, 6-gingerol is the most widely studied. Reviews describe it as anti-inflammatory, antioxidant, and anticancer in experimental models, with particular interest in gastrointestinal cancers because ginger compounds spend substantial time in the digestive tract.
That makes gingerol relevant not only as a food-based support compound, but also as a possible local-acting digestive tract agent in colorectal and gastric settings.
How Gingerol Works in Cancer
Pathways: NF-κB, COX-2, STAT3, MAPK, and PI3K/Akt/mTOR
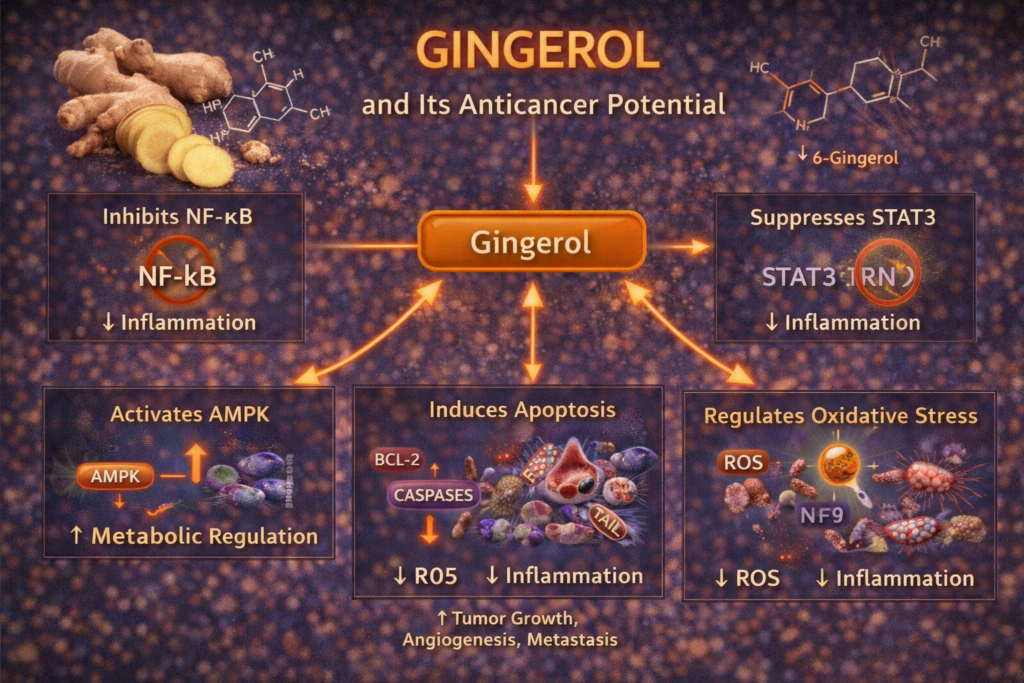
Gingerol affects several of the same pathways that appear throughout cancer biology. Reviews and experimental studies describe inhibition of NF-κB, COX-2, STAT3, MAPK signaling, AP-1 activity, Wnt/β-catenin, and PI3K/Akt/mTOR-related survival signaling. These are major systems tumors use for proliferation, inflammation, immune evasion, and spread.
That matters because cancer relies on networks, not isolated switches. By suppressing inflammatory signaling and growth pathways together, gingerol may make the tumor environment less favorable and less resilient. This connects naturally to:
https://helping4cancer.com/nf-kb-cancer/
https://helping4cancer.com/pi3k-akt-pathway-cancer/
Metabolism: Oxidative Stress, Glycolysis Pressure, and Mitochondria
Gingerol also connects to cancer metabolism. In healthy cells it can support antioxidant defenses, but in cancer cells it may raise reactive oxygen species enough to damage mitochondria and push the cell toward apoptosis. This selective pro-oxidant pressure is one reason researchers see gingerol as more than a general antioxidant. In pancreatic cancer models, ROS buildup and mitochondrial membrane disruption were linked to cancer cell death.
This places gingerol in the broader discussion around cancer metabolism, oxidative stress, and mitochondrial vulnerability:
https://helping4cancer.com/cancer-metabolism/
https://helping4cancer.com/oxidative-stress-cancer/
https://helping4cancer.com/redox-balance-cancer/
Immune System: Inflammation Control and Tumor Surveillance
Gingerol is not primarily known as a direct NK-cell stimulant like a beta-glucan mushroom, but it still matters to immune defense because it lowers inflammatory cytokines and may help create a less tumor-friendly environment. It also appears to support gut barrier function and microbiome balance, which indirectly affects immune surveillance. In digestive cancers, that gut-immune connection may be especially important.
For broader immune context, see:
https://helping4cancer.com/immune-system-cancer/
Apoptosis: Turning Cancer’s Self-Destruct Program Back On
One of gingerol’s strongest anticancer actions is apoptosis induction. Studies in colorectal and other cancer cell lines show activation of caspases 3, 7, 8, and 9, along with other changes associated with programmed cancer cell death. In colon cancer models, 6-gingerol has been shown to trigger phosphatidylserine externalization, a classic apoptosis marker, while sparing normal colon cells more effectively than cancer cells.
This matters because many tumors survive by blocking apoptosis. Gingerol may help reopen that pathway, making cells more likely to die instead of continuing to divide.
Oxidative Stress: Selectively Increasing ROS in Cancer Cells
Gingerol’s redox behavior is one of its most interesting features. In normal cells, it can act as an antioxidant. In cancer cells, it may increase ROS enough to create lethal stress. Reviews specifically describe this dual role as part of why gingerol can protect healthy tissues while still damaging tumor cells.
That selective ROS increase is especially relevant in:
- pancreatic cancer
- colorectal cancer
- chemotherapy-sensitization discussions
- metabolic and oxidative-stress strategies
NF-κB, COX-2, and STAT3: Cutting Off Survival Signals
NF-κB and STAT3 are two of cancer’s favorite survival systems. They help drive chronic inflammation, protect tumors from apoptosis, and support metastasis and treatment resistance. Gingerol has been shown to suppress NF-κB by preventing IκBα degradation and blocking p65 nuclear translocation in experimental models. It also downregulates COX-2 and inflammatory signaling linked to tumor promotion. Reviews further describe inhibition of STAT3 in cancer cells, helping reduce survival signaling and proliferation.
This makes gingerol especially relevant in inflammation-driven cancers and in any broader pathway-based strategy.
MAPK, AP-1, and Growth Control
Gingerol also modulates MAPK pathways such as ERK and JNK, which influence growth, stress response, and transcription factors like AP-1. In colon cancer cells, downregulation of these signals has been associated with lower proliferation and lower expression of tumor-promoting genes such as EGFR and COX-2.
This gives gingerol a broader anti-proliferative role than a single-pathway compound.
Wnt/β-Catenin and PI3K/Akt/mTOR: Slowing Tumor Progression
The Wnt/β-catenin pathway is central to colorectal cancer progression and stem-like behavior. Gingerol has been reported to reduce β-catenin-related signaling in experimental colorectal models. It also appears to inhibit PI3K/Akt/mTOR signaling, which helps tumors grow, survive, and adapt metabolically.
This is important because it connects gingerol to both tumor growth control and recurrence biology. For related reading:
https://helping4cancer.com/pi3k-akt-pathway-cancer/
https://helping4cancer.com/oncogenes-vs-tumor-suppressor-genes/
Anti-Angiogenesis and Metastasis Prevention
Gingerol may help reduce angiogenesis by lowering VEGF and MMP expression in preclinical models. That means fewer blood vessels feeding the tumor and less ability for cancer cells to break through tissues and spread. Reviews also describe anti-metastatic effects through lower invasion signaling and reduced EMT-related behavior.
This makes gingerol relevant to anti-angiogenic and anti-spread strategies, especially in digestive cancers and in combination settings.
Gingerol and Chemotherapy Synergy
One of the more practical areas of interest is combination therapy. A 2023 ovarian cancer study found that 6-gingerol plus cisplatin was more effective than either alone at inducing apoptosis and suppressing angiogenesis in experimental models. Reviews also discuss synergy with methotrexate and other agents in preclinical settings.
That does not prove clinical benefit in humans yet, but it does support gingerol’s role as a possible chemosensitizer rather than just a general health compound.
Gut Microbiome and Colorectal Cancer Support
Gingerol may also help by influencing the gut microbiome. Review literature describes improved microbial balance, increased beneficial bacteria such as Lactobacillus in colitis-associated models, stronger gut barrier function, and lower inflammatory cytokines. That is especially relevant in colorectal cancer, where chronic inflammation, dysbiosis, and barrier damage all increase risk.
This makes gingerol one of the more gut-smart compounds in the cancer-support space.
Role in Cancer Strategy
Gingerol fits best as a support and pressure compound rather than a stand-alone attack-phase tool.
Where It Fits Best
Gingerol may fit well in:
- support phases for digestive cancers
- meal-based use for gut-localized exposure
- inflammation-control strategies
- combination approaches with chemotherapy
- long-term metabolic and gut-health support
Why It Matters Strategically
Its main value comes from how it connects:
- apoptosis
- ROS pressure in tumors
- pathway suppression
- gut barrier support
- microbiome balance
- anti-angiogenesis
That makes it feel like part of a larger cancer system, not an isolated supplement.
Dosage and Safety
Human trials using about 2 grams per day of ginger extract have generally shown good tolerability, including in colorectal cancer prevention research. Pharmacokinetic work suggests free gingerols are present in plasma only briefly and much of the circulating material appears as conjugated metabolites, which is one reason bioavailability remains a challenge for systemic use.
A practical takeaway from your original page is reasonable:
- fresh ginger is useful as food support
- standardized extracts are more realistic for therapeutic-level exposure
- around 2 g/day of ginger extract has human safety data
- higher-impact systemic effects may depend on better formulations in the future
Key Benefits of Gingerol in Cancer Support
- promotes apoptosis in cancer cells
- increases ROS selectively in tumor cells
- suppresses NF-κB, COX-2, STAT3, MAPK, and PI3K/Akt/mTOR-related signaling
- may reduce angiogenesis through VEGF and MMP suppression
- supports gut barrier and microbiome balance
- may enhance chemotherapy response in preclinical models
- appears especially relevant in colorectal, gastric, pancreatic, and other GI cancers
Limits of the Evidence
The strongest evidence for gingerol is still preclinical. Most anticancer claims come from cell studies, animal models, mechanistic reviews, and early biomarker-based human work, not from large cancer-treatment trials. Human colorectal studies suggest safety and possible biomarker effects, but not proven cancer treatment outcomes.
So the most accurate position is:
gingerol is promising, biologically active, and especially relevant to digestive cancers, but it is not a proven stand-alone human cancer treatment.
Final Takeaway
Gingerol is one of the most interesting natural compounds in digestive-cancer support because it works across several important systems at once. It helps by:
- promoting apoptosis
- increasing oxidative stress inside cancer cells
- blocking inflammatory and survival pathways
- reducing angiogenesis and spread
- supporting the gut microbiome and intestinal barrier
That combination makes it especially valuable in a larger system-based strategy focused on inflammation, metabolism, gut health, and tumor pressure.
Related Topics
EMT and metastasis
https://helping4cancer.com/emt-cancer-metastasis/
The foundation of cancer
https://helping4cancer.com/the-foundation-of-cancer/
NF-κB and inflammatory cancer signaling
https://helping4cancer.com/nf-kb-cancer/
PI3K/Akt pathway and tumor survival
https://helping4cancer.com/pi3k-akt-pathway-cancer/
Cancer metabolism and tumor adaptation
https://helping4cancer.com/cancer-metabolism/
Oxidative stress and redox balance
https://helping4cancer.com/oxidative-stress-cancer/
https://helping4cancer.com/redox-balance-cancer/
Gut-linked cancer support and inflammation
https://helping4cancer.com/genomic-instability-cancer/
Gingerol and Its Anticancer Potential: Mechanisms, Pathways, and Dosage
Introduction to Gingerol
Gingerol, particularly [6]-gingerol, is the primary bioactive compound in fresh ginger (Zingiber officinale), contributing to its pungent flavor and medicinal properties. It has garnered attention for its anti-inflammatory, antioxidant, and anticancer effects, especially in gastrointestinal (GI) cancers like colorectal, gastric, pancreatic, and liver cancers, which are among the most prevalent and deadly globally. Ginger and Its Constituents: Role in GI Cancer
Anticancer Mechanisms Overview
Gingerol exerts anticancer effects by targeting cancer hallmarks such as uncontrolled proliferation, evasion of apoptosis, angiogenesis, and metastasis. Its selective toxicity to cancer cells while sparing normal cells makes it a promising chemopreventive agent. Studies show efficacy in colorectal, gastric, and pancreatic cancer models, primarily through apoptosis induction and inflammation suppression. Recent Updates on Ginger Bioactive Compounds
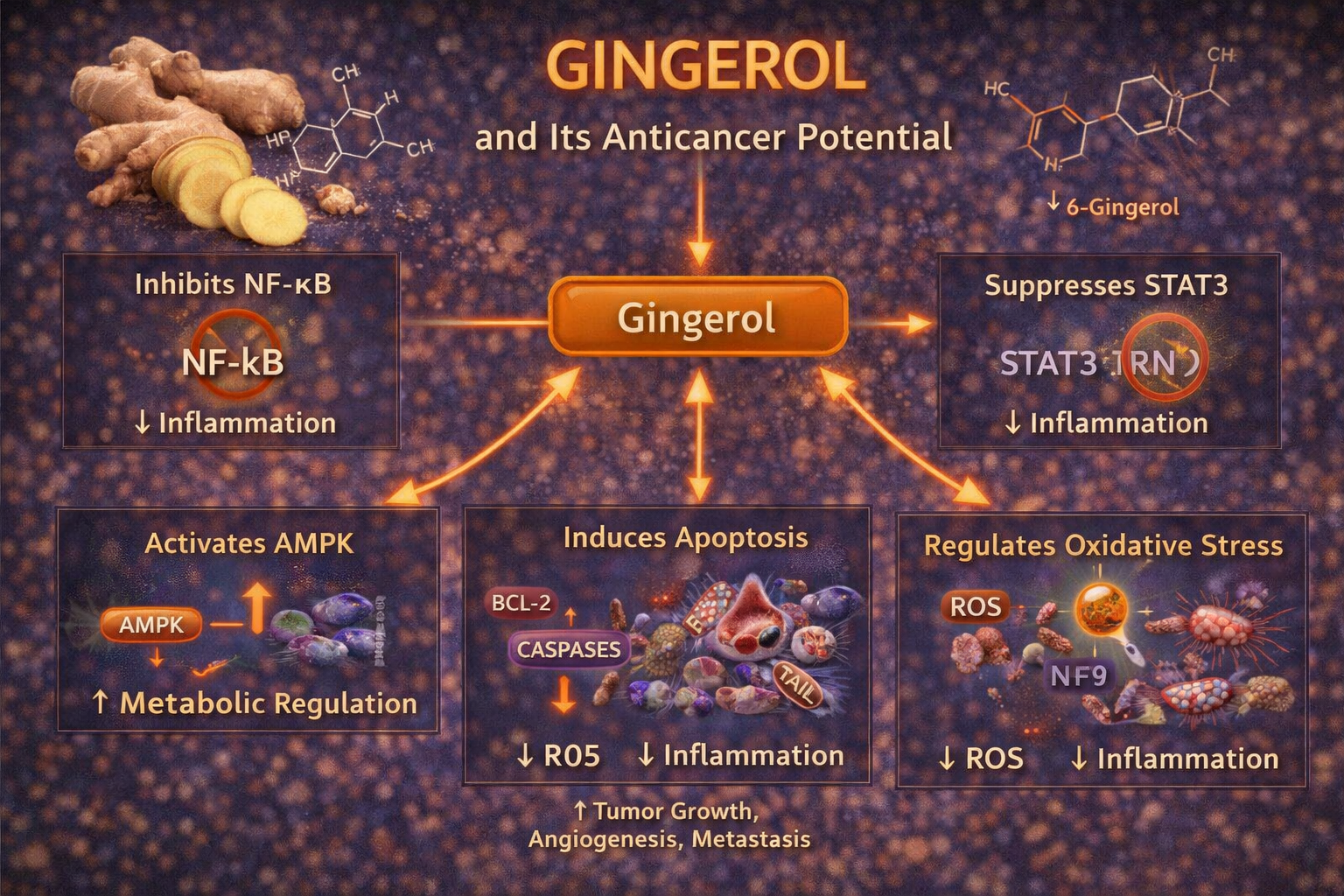
Apoptosis Induction
[6]-Gingerol induces apoptosis in cancer cells by activating caspases (8, 9, 3, and 7) and promoting PARP cleavage, as observed in colorectal cancer cells (SW-480). This caspase-dependent apoptosis involves phosphatidylserine externalization, a hallmark of programmed cell death, without harming normal colon cells. Ginger as an Anticolorectal Cancer Spice
Oxidative Stress-Mediated Apoptosis
Gingerol triggers apoptosis via oxidative stress by elevating reactive oxygen species (ROS) in cancer cells, disrupting mitochondrial membrane potential. In pancreatic cancer cells (PANC-1), [6]-gingerol increases ROS, leading to caspase-dependent apoptosis, while normal cells remain unaffected due to lower baseline ROS levels. Ginger and Its Constituents: Role in GI Cancer
NF-kB Pathway Inhibition
The NF-kB pathway, often constitutively active in cancers, promotes cell survival and angiogenesis. [6]-Gingerol inhibits NF-kB by preventing IκBα degradation and p65 nuclear translocation, reducing expression of pro-tumorigenic genes like VEGF and IL-8 in ovarian and gastric cancer cells. Recent Updates on Ginger Bioactive Compounds
COX-2 Suppression
Cyclooxygenase-2 (COX-2) drives inflammation and tumor growth through prostaglandin E2 (PGE2) production. [6]-Gingerol suppresses COX-2 expression by inhibiting p38 MAPK and NF-kB signaling, as seen in mouse skin models, reducing inflammation-driven tumorigenesis in GI cancers. Recent Updates on Ginger Bioactive Compounds
MAPK Pathway Modulation
Mitogen-activated protein kinase (MAPK) pathways (ERK1/2, JNK, p38) regulate proliferation and survival. [6]-Gingerol inhibits ERK1/2 and JNK phosphorylation in colorectal cancer cells (SW-480), reducing AP-1 activation and tumor growth. Ginger as an Anticolorectal Cancer Spice
AP-1 Inhibition
Activator protein-1 (AP-1), a transcription factor regulated by MAPK, promotes tumorigenesis. [6]-Gingerol downregulates AP-1 activity in colorectal cancer cells by inhibiting ERK1/2 and JNK, reducing expression of EGFR and COX-2. Ginger as an Anticolorectal Cancer Spice
STAT3 Pathway Suppression
STAT3 drives proliferation and survival in cancers. [6]-Gingerol inhibits STAT3 nuclear translocation in ovarian and pancreatic cancer cells, reducing cyclin D1 and Bcl-2 expression, leading to cell cycle arrest and apoptosis. Recent Updates on Ginger Bioactive Compounds
PI3K/Akt/mTOR Inhibition
The PI3K/Akt/mTOR pathway supports cancer cell survival. [6]-Gingerol downregulates phosphorylated PI3K and Akt, inhibiting tumor growth in colorectal and cervical cancers by reducing autophagy and promoting apoptosis. Ginger and Its Constituents: Role in GI Cancer
Wnt/β-Catenin Pathway
The Wnt/β-catenin pathway drives colorectal cancer progression. [6]-Gingerol reduces β-catenin, PKCε, and GSK-3β expression in HCT116 cells, inhibiting proliferation and metastasis. Ginger as an Anticolorectal Cancer Spice
EGFR Signaling
Epidermal growth factor receptor (EGFR) signaling promotes colorectal cancer growth. [8]-Gingerol inhibits EGFR, inducing cell cycle arrest and apoptosis by reducing PCNA and cyclin D1 expression. Recent Updates on Ginger Bioactive Compounds
Anti-Angiogenic Effects
Gingerol inhibits angiogenesis by suppressing VEGF and MMPs (regulated by NF-kB and AP-1) in pancreatic and ovarian cancer models, limiting tumor vascularization and metastasis. Recent Updates on Ginger Bioactive Compounds
Cell Cycle Arrest
[6]-Gingerol induces G0/G1 or G2/M cell cycle arrest in colorectal cancer cells (SW-480, LoVo) by upregulating p53 and p21, halting proliferation. This effect is sometimes caspase-independent, indicating diverse mechanisms. Ginger as an Anticolorectal Cancer Spice
Synergy with Chemotherapy
[6]-Gingerol enhances chemotherapeutic efficacy. In cervical cancer cells, it synergizes with cisplatin, increasing ROS and DNA damage, leading to G2/M arrest and apoptosis. Similar effects are seen with methotrexate in leukemia cells. 6-Gingerol and Cisplatin in Ovarian Cancer
Anti-Inflammatory Effects
Chronic inflammation fuels cancer. [6]-Gingerol reduces proinflammatory cytokines (TNF-α, IL-6) and mediators (iNOS, COX-2) in colitis models, creating an anti-tumorigenic environment. Ginger as an Anticolorectal Cancer Spice
Antioxidant Activity
[6]-Gingerol’s antioxidant properties inhibit xanthine oxidase and boost SOD and catalase, reducing oxidative stress in normal cells while selectively increasing ROS in cancer cells. Ginger and Its Constituents: Role in GI Cancer
Gut Microbiota Modulation
Gingerol modulates gut microbiota, reducing colorectal cancer risk. In DSS-induced colitis models, ginger increases beneficial bacteria (e.g., Lactobacillus murinus), restoring microbial balance and reducing inflammation. Ginger as an Anticolorectal Cancer Spice
Microbial Dysbiosis Correction
Dysbiosis promotes colorectal cancer. Gingerol corrects dysbiosis by enhancing beneficial bacteria and suppressing pathogens, improving intestinal barrier function and reducing inflammatory markers. Ginger as an Anticolorectal Cancer Spice
Bioavailability Challenges
Gingerol’s low to moderate bioavailability limits systemic effects, but its prolonged GI tract residence enhances efficacy against digestive cancers. Nanoformulations are being explored to improve absorption. Pharmacokinetics of Gingerols
Pharmacokinetics in Humans
In healthy subjects, [6]-gingerol reaches peak plasma concentrations (0.1-0.4 µg/mL) within 1-2 hours after 2 g ginger extract, sufficient for local GI effects but limited systemically. Pharmacokinetics of Gingerols
In Vitro Dosage
In vitro, [6]-gingerol inhibits colorectal cancer cell growth at 0.5 µmol/L, with stronger apoptosis at 30-100 µmol/L. Ginger juice (25 µL/mL) selectively targets cancer cells. 6-Gingerol Inhibits Colon Cancer Cells
Animal Model Dosage
In rats, [6]-gingerol at 2-5 mg/kg inhibits tumor growth without toxicity, suggesting a therapeutic range. Higher doses (25 mg/kg) reduce inflammation in sepsis models. Ginger and Its Constituents: Role in GI Cancer
Human Clinical Trials
Trials using 2 g/day ginger extract (~20 mg [6]-gingerol) for 28 days show safety and reduced COX-1 expression in colorectal cancer patients, but PGE2 results are inconsistent. Ginger: Is it Ready for Prime Time?
Dosage Per Pound
Animal studies (2-5 mg/kg) suggest 136-340 mg [6]-gingerol for a 150-pound (68 kg) person, or 0.9-2.27 mg/pound. Fresh ginger (~1 mg [6]-gingerol/100 g) is impractical at these doses, necessitating extracts. Ginger as an Anticolorectal Cancer Spice
Practical Dosage Estimation
A 2 g ginger extract (5% [6]-gingerol) provides ~100 mg [6]-gingerol, or 0.67 mg/pound for a 150-pound person, aligning with animal study doses. Clinical trials confirm safety at this level. Pharmacokinetics of Gingerols
Safety Profile
Gingerol is generally safe, with 2 g/day ginger extract causing minimal GI discomfort in trials. Its selective toxicity to cancer cells enhances its therapeutic potential. Ginger: Is it Ready for Prime Time?
Bioavailability Enhancement
Nanoformulations aim to overcome gingerol’s low systemic bioavailability, enhancing delivery to tumor sites and reducing required doses. Ginger and Its Active Compounds
Combination Therapies
Gingerol synergizes with cisplatin, gemcitabine, and methotrexate, enhancing ROS and apoptosis in cancer cells. Combinations with jujube polysaccharide or Gelam honey also show promise. 6-Gingerol and Cisplatin in Ovarian Cancer
Gut Microbiota and Cancer
By restoring microbial balance, gingerol reduces inflammation-driven colorectal cancer risk, enhancing beneficial bacteria and suppressing proinflammatory cytokines. Ginger as an Anticolorectal Cancer Spice
Oxidative Stress Balance
Gingerol’s dual role as an antioxidant in normal cells and pro-oxidant in cancer cells allows selective targeting, protecting healthy tissues while inducing cancer cell death. Ginger and Its Constituents: Role in GI Cancer
Clinical Trial Limitations
Small sample sizes and short durations limit clinical trial findings. While 2 g/day ginger extract is safe, its impact on cancer biomarkers requires further validation. Ginger: Is it Ready for Prime Time?
Future Research Directions
Future studies should focus on optimizing bioavailability, refining dosages, and exploring gingerol’s synergy with other therapies. Larger clinical trials are needed to confirm efficacy. Recent Updates on Ginger Bioactive Compounds
Novel Insights from Recent Studies
Recent research highlights gingerol’s role in stabilizing telomeric G-quadruplexes, potentially disrupting cancer cell replication. This opens new avenues for targeted therapies. Harnessing Gingerols for Telomeric RNA
Conclusion
[6]-Gingerol is a promising anticancer agent, particularly for GI cancers, by inhibiting NF-kB, COX-2, MAPK, STAT3, PI3K/Akt, and Wnt/β-catenin pathways, inducing apoptosis, and modulating gut microbiota. Doses of ~0.67 mg/pound (from 2 g extract) are safe and effective, but bioavailability challenges necessitate further research into delivery methods. Ginger and Its Constituents: Role in GI Cancer
![Ginger root with [6]-gingerol molecules targeting cancer cells, inhibiting NF-kB and COX-2 for digestive cancer prevention.](https://helping4cancer.com/wp-content/uploads/2025/06/Learn-More-About-Gingerol-Anticancer-Benefits-1024x401.jpg)
Table of Contents